Tuberous Sclerosis Complex
A Timeline for Success: The FDA Approves New Topical Treatment for Facial Tumors in TSC



Posted May 18, 2022
Mary Kay Koenig, M.D., UTHealth Houston
 Dr. Mary Kay Koenig
Dr. Mary Kay Koenig
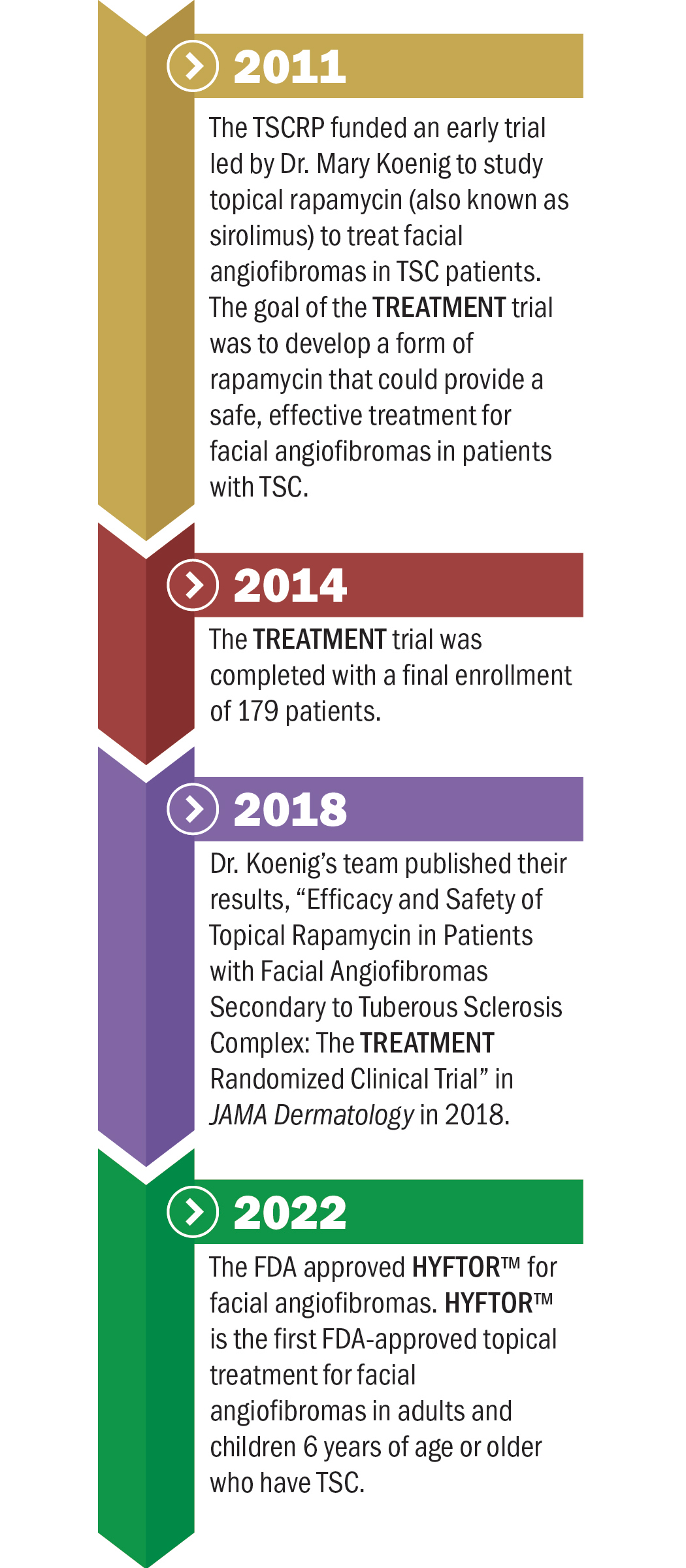
The recent U.S. Food and Drug Administration (FDA) approval of HYFTORTM to treat facial angiofibromas associated with tuberous sclerosis complex (TSC) was great news to the TSC community, as there was previously no effective permanent treatment for this condition. The journey of developing a similar drug began over a decade ago with a clinical trial funded by the Tuberous Sclerosis Complex Research Program (TSCRP).
TSC affects 1 in 6,000 individuals worldwide and 50,000 individuals in the United States. TSC is a genetic disorder characterized by abnormal skin pigmentation and tumor formation in multiple organ systems. The genes associated with TSC are involved in controlling cell growth. Common symptoms of TSC include learning disabilities, seizures, skin lesions, kidney tumors, lung disease, heart tumors, and brain tumors. Facial angiofibromas are benign skin tumors on the face, which are present in up to 80% of TSC patients. These facial lesions create considerable cosmetic morbidity for patients with TSC. Although there are surgical procedures available to remove the tumors, the majority of these treatments are uncomfortable and lack long-term efficacy.
In 2009, Dr. Mary Kay Koenig, M.D., with McGovern Medical School at UTHealth Houston, conducted the first small clinical trial using a topical rapamycin cream to treat cutaneous manifestations in TSC and neurofibromatosis I patients. Based on the preliminary results, in 2010, Dr. Koenig received a TSCRP Clinical Research Award to pursue a phase 2 randomized clinical trial to study the safety and efficacy of topical rapamycin to treat TSC-related facial angiofibromas (named the TREATMENT trial). The TREATMENT trial started in 2011. Patients were randomized at 1:1:1 ratio to 1% rapamycin, 0.1% rapamycin, or a control. The topical formulation was applied once to designated areas at bedtime for 6 months. The outcome measures included the angiofibromas grading scale (AGS) at baseline and 6 months by independent, masked dermatologists. The safety analysis included adverse events and serum rapamycin levels. In 2014, the trial was successfully completed, enrolling 179 patients over 10 sites. At 6 months, the rapamycin treatment groups had significant AGS improvement compared to the control group. AGS results were improved by 16.7 points within the 1% rapamycin group and 11.2 points in the 0.1% rapamycin group, while the control group only had 2.1 points of improvement. Topical rapamycin was generally well-tolerated with no measurable systemic absorption. The positive outcomes from the phase 2 trial laid the foundation for the next step of the drug development. In 2018, Dr. Koenig published the phase 2 clinical trial in JAMA Dermatology1 which demonstrated the safety and efficacy of the topical formulation of rapamycin in the treatment of TSC-related facial angiofibromas.
Dr. Koenig remarks, "Most people with TSC will develop facial angiofibromas. It can be very stigmatizing, disfiguring, and embarrassing." Previously there was no effective method for treating this condition. Of all the hard work and dedication that her team has shown over the lengthy process of bringing a therapeutic from bench top to bedside, Dr. Koenig writes, "I have dedicated my life and my career to taking care of people with rare diseases. Everybody knows someone who has a rare disease. Collectively they are not rare."
References:
1Koenig MK, Bell CS, Hebert AA, et al. 2018. JAMA Dermatology 154(7):773-780.
Links:
Last updated Monday, September 8, 2025
 An official website of the United States government
An official website of the United States government
 ) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
