Tuberous Sclerosis Complex
Autophagy and the Neurocognitive Deficits in TSC



Posted May 6, 2022
Guomei Tang, Ph.D., Department of Neurology, Columbia University Medical Center
 Dr. Guomei Tang
Dr. Guomei Tang
Tuberous sclerosis complex (TSC) is a multi-system genetic disorder caused by mutations in either the TSC1 or TSC2 gene. Cognitive impairments, including long-term and working memory deficits, are common neuropsychiatric features of a majority of TSC patients.
Recent evidence suggests that disinhibition of a specific protein kinase, mechanistic target of rapamycin (mTOR), contributes to TSC-related learning and memory deficits. The mTOR inhibitor rapamycin corrects both synaptic and cognitive defects in Tsc1 and Tsc2 heterozygous mutant mice. Clinical trials are thus underway to test the effects of rapamycin analogues, e.g., Everolimus, on neurocognitive problems in TSC patients. One caveat for rapamycin treatment is that long-term therapy is required to maintain effectiveness, which may cause adverse side effects.
In work funded by a Fiscal Year 2015 (FY15) Tuberous Sclerosis Complex Research Program (TSCRP) Idea Development Award, Dr. Guomei Tang at the Columbia University Medical Center investigated the role of impaired mTOR-dependent autophagy in TSC associated neurocognitive impairments. The main goals of this study were to determine the role of impaired mTOR-autophagy in cognitive dysfunction in Tsc1 or Tsc2-mutant mice, identify synaptic mechanisms of impaired mTOR-autophagy for cognitive dysfunction, and identify molecular mechanisms of impaired autophagy for synaptic dysfunction. Dr. Tang's research team found that the induction of autophagy was suppressed in Tsc1/2 mutant mice. Within the brain region responsible for learning and memory (the hippocampus), synapse maturation and long-term synaptic plasticity were impaired in autophagy-deficient mice. Further, as the mice aged, social and cognitive deficits became apparent in autophagy-deficient mice. Inhibiting mTOR by rapamycin corrects cognitive deficits in Tsc1/2-mutant mice but not in mice deficient for both Tsc1/2 and autophagy. Study results strongly suggested that autophagy is a promising therapeutic target for TSC-associated neuropsychiatric symptoms.
Dr. Tang will follow up this successful FY15 project with a new FY21 TSCRP Idea Development Award to investigate autophagy induction as a novel therapeutic strategy for TSC-associated cognitive and autistic social deficits. The goal of this new project is to develop a novel alternative treatment paradigm for TSC patients. The researchers will assess the safety and therapeutic efficacy of two well-characterized pharmacologic agents that are capable of augmenting autophagy in vivo. Dr. Tang hypothesizes that inducing autophagy will be effective in rescuing TSC-associated social and cognitive deficits. This work could set the stage for novel therapeutics. If both preclinical and clinical studies confirm that autophagy induction is an effective intervention for TSC-associated neuropsychiatric symptoms, the findings could rapidly translate into new clinical practices.
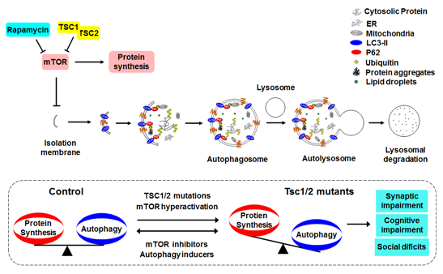 Figure 1: mTOR regulated autophagy and neurocognitive deficits in TSC. Autophagy is a lysosome dependent degradation pathway for the removal of unwanted material, e.g., excess or damaged proteins and organelles, inside living cells. The process of autophagy begins with the formation of cup-shaped isolation membranes, which engulf a portion of cytoplasm and enclose into double membrane autophagosomes. Autophagosomes are transported to and fuse with lysosomes, in which the sequestered cargoes are degraded. Autophagy is negatively regulated by mTOR kinase. TSC1/2 mutations disinhibit mTOR, leading to excessive protein synthesis and impaired autophagy, and as a consequence, synaptic impairment and TSC associated cognitive and social deficits. Inhibiting mTOR and activating autophagy can both re-establish a balance between protein synthesis and autophagy, and correct synaptic, cognitive and social deficits associated with Tsc1/2 mutations.
Figure 1: mTOR regulated autophagy and neurocognitive deficits in TSC. Autophagy is a lysosome dependent degradation pathway for the removal of unwanted material, e.g., excess or damaged proteins and organelles, inside living cells. The process of autophagy begins with the formation of cup-shaped isolation membranes, which engulf a portion of cytoplasm and enclose into double membrane autophagosomes. Autophagosomes are transported to and fuse with lysosomes, in which the sequestered cargoes are degraded. Autophagy is negatively regulated by mTOR kinase. TSC1/2 mutations disinhibit mTOR, leading to excessive protein synthesis and impaired autophagy, and as a consequence, synaptic impairment and TSC associated cognitive and social deficits. Inhibiting mTOR and activating autophagy can both re-establish a balance between protein synthesis and autophagy, and correct synaptic, cognitive and social deficits associated with Tsc1/2 mutations.
Links:
Last updated Monday, September 8, 2025
 An official website of the United States government
An official website of the United States government
 ) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
