- Using a Drosophila Model to Screen for Molecules to Reverse the Effects of Tsc Gene Mutations
- Role of Altered mGluR Activity in Cognitive Impairments in TSC: Implications for a Novel Method of Treatment
- LAMS Clinical Research Network
- Understanding the Etiology of Tuberous Sclerosis Complex
Using a Drosophila Model to Screen for Molecules to Reverse the Effects of Tsc Gene Mutations
Posted October 16, 2012
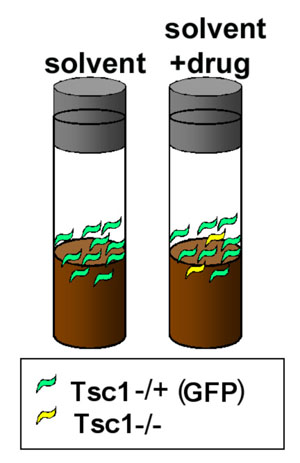
Tin Tin Su, Ph.D., University of Colorado, Boulder, ColoradoTsc1 and Tsc2 normally function to suppress cellular growth. Tuberous sclerosis complex (TSC) results from mutation of the Tsc1 or Tsc2 genes, causing excessive cell growth and tumor formation. Dr. Tin Tin Su, with funding from a Fiscal Year 2005 (FY05) Tuberous Sclerosis Complex Research Program (TSCRP) Concept Award, previously developed a Drosophila melanogaster (fruit fly) model to screen for small molecules that can reverse the effects of Tsc1/2 mutations. Through funding from an FY09 TSCRP Exploration - Hypothesis Development Award, Dr. Su's research group utilized this Drosophila model to screen approximately 6,000 small molecules. This work led to the identification of two small molecules that reliably inhibit the effects of mutant Tsc1 in Drosophila. The next steps in this work will involve validating the activity of these candidate small molecules using the Drosophila model and characterizing their mechanism of action.
Schematic of Drosophila melanogaster (fruit fly) model to screen for small molecules that can reverse the effects of Tsc1/2 mutations.
Links:
Role of Altered mGluR Activity in Cognitive Impairments in TSC: Implications for a Novel Method of Treatment
Posted August 16, 2012
Mark Bear, Ph.D., Massachusetts Institute of Technology
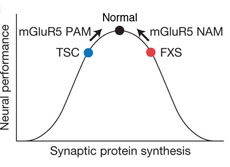
Patients with Tuberous Sclerosis Complex (TSC) develop benign tumors and suffer from cognitive deficiencies including mental retardation, epilepsy, autism, anxiety, and mental disorders. TSC is a genetic disorder resulting from the mutation of either the TSC1 or TSC2 gene. In healthy cells, the TSC proteins function to modulate protein synthesis by suppressing mTOR (mammalian target of rapamycin) signaling. Interestingly, maintenance of synaptic plasticity, critical for learning, memory, and cognition, requires precise regulation of protein synthesis. Based on these observations, Dr. Mark Bear hypothesized that dysregulation of synaptic protein synthesis may contribute to TSC-associated learning and cognitive deficiencies. Dr. Bear's research team, with funding from an FY10 TSCRP Idea Development Award, demonstrated that loss of TSC2 resulted in unregulated mTOR activity and suppressed hippocampal protein synthesis. In addition, the team also determined that treatment with an mGluR5 positive allosteric modulator (PAM) restored hippocampal protein synthesis. Additionally, treatment with mGluR5 PAM also reversed hippocampal-dependent behavioral deficits in Tsc2+/- mice. These exciting results suggest that mGluR5 PAM may be a novel therapeutic intervention for TSC-associated cognitive deficiencies.
Link:
LAMS Clinical Research Network
Posted May 15, 2012
Francis McCormack, M.D., University of Cincinnati
Tuberous sclerosis complex (TSC) is a rare genetic disorder, affecting approximately one million people worldwide, that may result in seizures, cognitive impairment, and benign tumors of the skin, eyes, brain, and other organs. Lymphangioleiomyomatosis (LAM), a life-threatening progressive lung manifestation of TSC, affects approximately 40 percent of women with TSC. Although a significant number of molecular targets have been identified as potential therapeutics for LAM, the design and implementation of clinical trials has been limited due to the rare occurrence of the disease. Dr. Francis McCormack at the University of Cincinnati received a fiscal year 2009 Clinical and Translational Research Award from the TSC Research Program to develop a novel paradigm for conducting clinical trials in rare diseases more quickly and more conveniently to patients by bringing clinical trials to local LAM treatment sites. To this end, Dr. McCormack established the LAM Clinical Research Network (LCRN), a collaboration of 24 LAM clinics throughout the United States. LAM clinic directors are in the process of developing a longitudinal dataset that would be collected from all consenting participants to build a natural history database and maintain a list of prospective subjects for future clinical trials. The LCRN will allow patients to enroll in studies at their local LAM clinics with their physician, rather than traveling, sometimes long distances, to gain entry into cutting-edge clinical trials.
One of the initial LCRN aims is to develop and validate a biomarker for diagnosis of LAM using vascular endothelial growth factor-D (VEGF-D). Preliminary results suggest that serum VEGF-D levels can be utilized to diagnosis LAM in women with TSC. A second project of the LCRN is a Phase I/II trial of Letrozole, an oral non-steroidal aromatase inhibitor, in postmenopausal women with LAM (Trial of Aromatase Inhibition in LAM, or TRAIL), which is expected to begin enrollment at six LCRN clinics in 2012.
Dr. McCormack and the LCRN are in a unique position to increase access to clinical trials for individuals with LAM and, in turn, facilitate the delivery of novel LAM treatment options to patients.Link:
Public and Technical Abstracts: LAMS Clinics Research Network
Understanding the Etiology of Tuberous Sclerosis Complex
Posted April 10, 2012
Dr. Angelique Bordey, Yale University
Tuberous sclerosis complex (TSC) patients have mutations in the Tsc1 or Tsc2 genes and suffer from the formation of lesions on multiple organs. Patients also develop neurological symptoms including seizures, mental retardation, and autism. Cortical tuber lesions that form during embryonic development are associated with the development of seizures in TSC patients. The mechanisms involved in TSC lesion formation and seizure generation are not well understood and validated animal models to investigate the etiology or cause of TSC lesions and subsequent development of neurological disorders are currently unavailable. Through funding from a fiscal year 2009 Idea Development Award, Dr. Angelique Bordey, at Yale University, has developed and validated a mouse model to bridge this gap in knowledge. Dr. Bordey and her research team have determined that deletion of Tsc1 in this mouse model leads to formation of lesions that contain many of the hallmark characteristics associated with human cortical tubers in TSC. This animal model will now allow TSC researchers to study the development of cortical tuber lesions at specific time points during embryonic development. This work was recently published in the Journal of Clinical Investigation (Feliciano 2011).
Link:
Public and Technical Abstracts: Understanding the Etiology of Tuberous Sclerosis Complex