Tuberous Sclerosis Complex
Identifying the Roots of Myelin Dysfunction in the TSC Brain
Posted March 10, 2017
Mustafa Sahin, M.D., Ph.D., Boston Children's Hospital

Photo provided courtesy of Boston Children's Hospital
Tuberous sclerosis complex (TSC) is an autosomal dominant disorder caused by a mutation in either of the TSC1 or TSC2 genes. Neurological symptoms are common and include presentations with autism, developmental delays, and epilepsy. These symptoms are attributed to two key features of the disease: hamartomas, or cortical tubers in the brain, and hypomyelination. Hypomyelination occurs when glial cells in the brain are unable to efficiently generate the insulating fatty myelin sheath which surrounds nerve fibers and facilitates rapid and efficient transmission of nerve impulses. Recent work from the lab of Dr. Mustafa Sahin, at Boston Children's Hospital, has led to a better understanding of the cause of this decreased myelination in TSC brains.
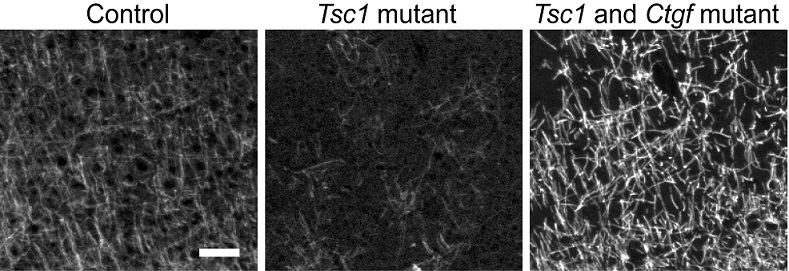
Dr. Sahin's lab discovered that neurons in TSC brains expressed an increased amount of a protein known as Connective Tissue Growth Factor (CTGF) compared to normal cells. CTGF, which is secreted from the neurons to the extracellular space, acts on oligodendrocytes-glial cells in the brain that support neuronal function and help build the myelin sheath structure and prevents the normal myelination process. With funding from a fiscal year 2012 TSCRP Idea Development Award, Dr. Sahin investigated the role of CTGF in hypomyelination, and the role it may play in the TSC brain. Through these experiments, Dr. Sahin's team found that the genetic deletion of CTGF in neuronal cells that also lack TSC1, resulted in improvement of the ability of oligodendrocytes to produce myelin and develop normally.
When the team investigated CTGF expression in human neurons derived from fibroblasts of TSC patients and controls, they were able to confirm increased expression in the TSC affected neurons, confirming the link between mouse models and human. Once this connection was established, the team looked into the modes by which TSC-deficiencies might impact CTGF expression. They found that the protein serum response factor, SRF, which has previously been found to repress transcription of the CTGF gene, is expressed at decreased levels in neurons that are lacking TSC2 gene expression and in TSC1 mouse model brains. This suggests that CTGF upregulation, and the resultant hypomyelination in TSC brains may be a result of the downregulation of SRF.
Dr. Sahin and his team plan to continue their investigations into the effects of CTGF on myelination, and the mechanism by which CTGF expression can impact the brain. The hope is that by understanding the causes and pathways that lead to these neurological phenotypes and impairments, it will be possible to develop therapies that could block the impact of CTGF expression and restore myelination to normal levels. Not only would such a therapy have an impact on TSC patients, but on patients suffering from similar myelination deficit disorders, including multiple sclerosis and cerebral palsy.
Reference:
Ebru Ercan, Juliette M. Han, Alessia Di Nardo, Kellen Winden, Min-Joon Han, Leonie Hoyo, Afshin Saffari, Andrew Leask, Daniel H. Geschwind, Mustafa Sahin. Neuronal CTGF/CCN2 negatively regulates myelination in a mouse model of tuberous sclerosis complex. The Journal of Experimental Medicine, 2017; jem.20160446 DOI: 10.1084/jem.20160446
Link:
Public and Technical Abstracts: Role of CTGF in White Matter Development in Tuberous Sclerosis
Last updated Wednesday, March 12, 2025














