Tuberous Sclerosis Complex
TSC-FoxO Signaling Network in Kidney Cancer Development
Posted March 31, 2016
Boyi Gan, Ph.D., University of Texas MD Anderson Cancer Center
 Tuberous Sclerosis Complex (TSC) is caused by a mutation in either the TSC1 or TSC2 gene and is characterized by the formation of non-malignant tumors in a variety of organs, including kidney, brain, heart, and lung. Common complications in individuals with TSC include seizures, developmental delay, intellectual disability, and autism. However, the most frequent cause of death related to TSC stems from complications of tumors in the kidneys.
Tuberous Sclerosis Complex (TSC) is caused by a mutation in either the TSC1 or TSC2 gene and is characterized by the formation of non-malignant tumors in a variety of organs, including kidney, brain, heart, and lung. Common complications in individuals with TSC include seizures, developmental delay, intellectual disability, and autism. However, the most frequent cause of death related to TSC stems from complications of tumors in the kidneys.
Funded by a Fiscal Year 2009 (FY09) Career Transition Award from the Tuberous Sclerosis Complex Research Program (TSCRP), Dr. Boyi Gan, originally at Dana-Farber Cancer Institute and currently at the University of Texas MD Anderson Cancer Center, sought to elucidate the molecular mechanisms behind the development of TSC-related renal tumors.
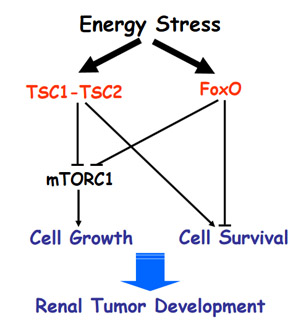
The mammalian target of rapamycin complex 1 (mTORC1) is a key regulator of protein synthesis and cell growth and plays a key role in the development of many human cancers, including renal cancers. Therefore, further elucidating the mTORC1 signaling and its role in the development of cancers could provide valuable insight into potential treatments for renal cancers. In this study, Dr. Gan sought to test the hypothesis that the transcription factor forkhead box O (FoxO) plays a key role in the TSC-mediated tumorigenesis via the mTORC1 pathway. Dr. Gan showed that FoxO cooperates with the TSC1-TSC2 complex to inhibit mTORC1 signaling under energy stress conditions, and inactivation of FoxO reduces the repression of mTORC1. In addition, he showed that, in response to energy stress, FoxO upregulates the expression of BCL2/adenovirus E1B 19 kDa protein-interacting protein 3 (BNIP3); BNIP3 then binds to and inhibits Rbeb, leading to mTORC1 inactivation in an AMPK-TSC-independent manner. Further validating these findings in human renal cancers, he showed downregulation of the FoxO-BNIP3 signaling axis, which was associated with poor clinical outcomes.
Dr. Gan's research, supported by the TSCRP, has provided significant insight into the role of FoxO in the TSC-mTOR signaling pathway in renal tumorigenesis, which may ultimately help in the development of novel drugs against renal complications in TSC patients.
The TSCRP Career Transition Award has also helped Dr. Gan advance his career as a researcher. He was able to transition from a mentored position at the Dana-Farber Cancer Institute to an independent faculty position, and, with the help of data obtained as a result of his TSCRP-funded work, has received additional funding to continue his TSC research, including grants from the National Institutes of Health. Dr. Gan is currently an Assistant Professor, with his own research team at the MD Anderson Cancer Center, where he continues his research to investigate the mechanisms behind TSC, with the goal of providing more insight into therapies for TSC and other cancers.
Publications:
Liu X, Xiao Z, Han L, et al. 2016. LncRNA NBR2 engages a metabolic checkpoint by regulating AMPK under energy stress. Nature Cell Biol (In press).
Lin A, Piao H, Zhuang L, et al. 2014. FoxO transcription factors promote AKT Ser473 phosphorylation and renal tumor growth in response to pharmacological inhibition of the PI3K-AKT pathway. Cancer Res 74(6):1682-1693.
Lin A, Yao J, Zhuang L, et al. 2014. FoxO-BNIP3 axis exerts a unique regulation of mTORC1 and cell survival under energy stress. Oncogene 33(24):3183-3194.
Gan B, Lim C, Chu G, et al. 2010. FoxOs enforce a progression checkpoint to constrain mTORC1- activated renal tumorigenesis. Cancer Cell 18 (5):472-484.
Links:
Public and Technical Abstracts: TSC-FoxO Signaling Network in Kidney Cancer Development














