Posted September 15, 2015
Reuben J. Shaw, Ph.D., The Salk Institute for Biological Studies
 Autophagy is a cellular process responsible for the destruction of unnecessary or damaged components of a cell. This process has a key role in the cell's response to nutrient-deprived conditions and other stressors by breaking down those components to their basic elements to serve as a nutrient source to maintain normal cellular function. Some cancers have the ability to upregulate this process in order to survive the cellular stress that is produced by the tumor environment, thereby allowing the tumor to increase its growth and aggressiveness. This suggests that the inhibition of autophagy in the tumor may be advantageous for the design of cancer therapy regimens.
Autophagy is a cellular process responsible for the destruction of unnecessary or damaged components of a cell. This process has a key role in the cell's response to nutrient-deprived conditions and other stressors by breaking down those components to their basic elements to serve as a nutrient source to maintain normal cellular function. Some cancers have the ability to upregulate this process in order to survive the cellular stress that is produced by the tumor environment, thereby allowing the tumor to increase its growth and aggressiveness. This suggests that the inhibition of autophagy in the tumor may be advantageous for the design of cancer therapy regimens.
Dr. Reuben J. Shaw received funding from a Fiscal Year 2012 Tuberous Sclerosis Complex Research Program (TSCRP) Idea Development Award to investigate whether inhibition of a protein in the autophagy pathway, ULK1, has therapeutic potential for tuberous sclerosis complex (TSC). TSC is a rare genetic disease with many manifestations, including the formation of widespread benign tumors predominantly in the brain, skin, kidneys, heart, and lung. TSC is characterized by mutations in the TSC1 or TSC2 gene that cause increased activation of the mammalian target of rapamycin (mTOR) signaling pathway. The mTOR pathway is involved in cell growth, metabolism, and proliferation; when upregulated in TSC, it causes abnormal cell growth leading to TSC tumors. mTOR inhibitors, which were shown to suppress the elevated mTOR found in TSC patient cells, are used currently to treat TSC. Cells and tumors with hyperactive mTOR signaling - such as those containing TSC mutations - have been shown to have a suppressed ULK1 complex and autophagy process. As the effects of many mTOR inhibitors can be cytostatic, inducing growth arrest, but not killing of tumor cells, Dr. Shaw hypothesized that the induction of ULK1 and autophagy by mTOR inhibitors may be allowing them to survive. Dr. Shaw therefore hypothesized that inhibiting both mTOR and ULK1 simultaneously would provide a potent combined treatment option for those with TSC.
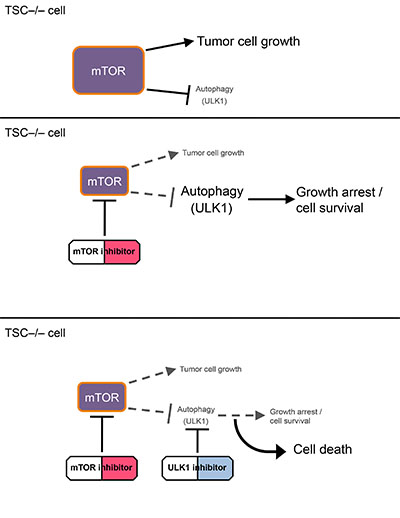 Dr. Shaw has recently published findings from his TSCRP-funded research, which newly identifies and characterizes a small molecule, SBI-0206965, that selectively inhibits ULK1. Dr. Shaw's team shows that SBI-0206965 suppresses autophagy induced by mTOR inhibition and prevents ULK1-dependent cell survival following nutrient deprivation by enhancing apoptosis in tumor cells. SBI-0206965 acts as a target inhibitor of ULK1 by converting the growth arrest of cells into a cell death response. This causes the cells to then lose their autophagic maintenance of cell survival, making SBI-0206965 a valuable tool for future autophagy studies. The results of this research also provide early preclinical data to support further development of SBI-0206965 or an analog (structurally similar molecule) for investigation as a therapeutic for TSC.
Dr. Shaw has recently published findings from his TSCRP-funded research, which newly identifies and characterizes a small molecule, SBI-0206965, that selectively inhibits ULK1. Dr. Shaw's team shows that SBI-0206965 suppresses autophagy induced by mTOR inhibition and prevents ULK1-dependent cell survival following nutrient deprivation by enhancing apoptosis in tumor cells. SBI-0206965 acts as a target inhibitor of ULK1 by converting the growth arrest of cells into a cell death response. This causes the cells to then lose their autophagic maintenance of cell survival, making SBI-0206965 a valuable tool for future autophagy studies. The results of this research also provide early preclinical data to support further development of SBI-0206965 or an analog (structurally similar molecule) for investigation as a therapeutic for TSC.
There are a number of ongoing clinical trials that combine chloroquine, a general autophagy inhibitor, or its analogs, with existing cancer therapies, including mTOR inhibitors. Dr. Shaw's data show that SBI-0206965 is able to induce more cell death than chlorquine when combined with mTOR inhibitors in cultured cells. By combining a ULK1 inhibitor such as SBI-0206965 with mTOR inhibitors as a treatment option, this may allow for lower dosing schedules of mTOR inhibitors and help to avoid therapeutic resistance for those patients currently treated with mTOR inhibitors. With further development of SBI-0206965 and future research in animal models and clinical trials, SBI-0206965 may one day provide a powerful drug therapy option for the treatment of TSC tumors.
Publications:
Egan DF, Chun MG, Vamos H, et al. 2015. Small molecule inhibition of the autophagy kinase ULK1 and identification of ULK1 substrates. Molecular Cell 59(2):285-297.
Links:














