Posted July 14, 2015
Kun-Liang Guan, Ph.D., University of California, San Diego
 Proteins regulate many signaling pathways in the body by upregulating and downregulating certain molecules and/or enzymes so that the needs of the cell are met. The mammalian target of rapamycin (mTOR) signaling pathway is involved in cell growth, metabolism, and proliferation. A wide range of extracellular and intracellular signals regulate this pathway, including TSC1 and TSC2 proteins. Mutations in either TSC1 or TSC2 can have a major impact on mTOR signaling. Tuberous sclerosis complex (TSC) is a genetic disorder caused by mutations in the TSC1 or TSC2 tumor suppressor gene and is characterized by seizures, developmental delays, kidney disease, behavioral problems, and the growth of benign tumors in many organs. Determining the role of TSC1/2 in mTOR signaling has been paramount in understanding TSC disease and in identifying targets for new therapeutics.
Proteins regulate many signaling pathways in the body by upregulating and downregulating certain molecules and/or enzymes so that the needs of the cell are met. The mammalian target of rapamycin (mTOR) signaling pathway is involved in cell growth, metabolism, and proliferation. A wide range of extracellular and intracellular signals regulate this pathway, including TSC1 and TSC2 proteins. Mutations in either TSC1 or TSC2 can have a major impact on mTOR signaling. Tuberous sclerosis complex (TSC) is a genetic disorder caused by mutations in the TSC1 or TSC2 tumor suppressor gene and is characterized by seizures, developmental delays, kidney disease, behavioral problems, and the growth of benign tumors in many organs. Determining the role of TSC1/2 in mTOR signaling has been paramount in understanding TSC disease and in identifying targets for new therapeutics.
Dr. Kun-Liang Guan of the University of California, San Diego, and formally of the University of Michigan, has provided valuable insight into the role of the mTOR pathway in TSC. Over a span of a decade, Dr. Guan has received six awards from the Tuberous Sclerosis Complex Research Program (TSCRP) to study this relationship. The results of his research have significantly advanced the TSC field and have helped identify potential drug targets for the treatment of TSC, including the demonstration of TSC1 and TSC2 as the key upstream regulators of mTOR.
With funding from a Fiscal Year 2004 (FY04) TSCRP Concept Award, Dr. Guan showed that Herc1 positively regulates mTOR activity by promoting the degradation of TSC2. His research also suggested that TSC1 prevented this degradation by disrupting the interaction between TSC2 and Herc1. This meant that TSC2 was protected from Herc1-mediated degradation when TSC1 was in complex with TSC2. This demonstrated how functional TSC proteins worked together to affect mTOR signaling and suggested a consequence when one of these proteins was rendered inactive by mutation, as can be the case in TSC disease.
Dr. Guan received a FY05 TSCRP Concept Award to further investigate regulators of the mTOR pathway and discovered that TSC1 regulates TOR complex 2 (TORC2) indirectly through the TORC1/S6k pathway. mTOR is well known to exist in two distinct complexes, TORC1 and TORC2. At the time of the award, few scientific discoveries had been made in the functional importance and regulation of TORC2. Dr. Guan's results were among the first to demonstrate that TSC1 regulates TORC2 function. This discovery catapulted an entire subcategory of mTOR research aimed at understanding the role and mechanisms by which TSC1 regulates TORC2.
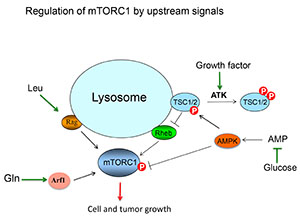
Dr. Guan was awarded another TSCRP Concept Award in FY05 to test his hypothesis that activation of p53 by mTOR and AMPK is the underling mechanism for energy-starvation induced programmed cell death in TSC-deficient cells. AMPK is a cellular energy sensor that is activated by energy starvation and phosphorylates TSC2 to inhibit the mTOR pathway and prevent unregulated cell growth. Dr. Guan was able to demonstrate that AMPK contributes to p53 stabilization, which he also showed is required for energy-starvation induced programmed cell death in cells lacking functional TSC2 proteins. Results from the study also provided evidence that p53 activity is dramatically increased in these cells, those lacking a functional TSC1 gene, as well as angiomyolipomas, non-malignant growths usually found in the kidneys of most individuals with TSC disease. Taken together, these results explained the sensitivity of TSC-deficient cells to energy starvation that can lead to misregulated p53 activation, causing the increased likelihood of tumor growth in patients with TSC disease.
With funding received from a FY06 TSCRP Concept Award, Dr. Guan investigated the role of Bnip3 in regulating cell growth in hypoxic (low oxygen) conditions. While it was well known that hypoxic conditions inhibit mTOR, the mechanism was not well understood. Dr. Guan's results demonstrated that Bnip3 directly binds Rheb, a protein that he and others have known to activate TORC1, and inhibits its ability to activate TORC1 and also mediates the inhibitory effect of hypoxia on mTOR signaling. These data identified Bnip3 as a tumor suppressor involved in inhibiting the mTOR pathway, thereby inhibiting cell growth, in hypoxic conditions.
Dr. Guan's research has led to many breakthroughs in the field of mTOR signaling, its involvement with TSC proteins, and its crosstalk with other well-known signaling mediators, as noted above. By 2012, and with funding from a FY08 TSCRP Idea Development Award, Dr. Guan had identified the mechanism by which Rag GTPase regulates TORC1 activation, additional regulators of TORC1 in response to amino acids (protein building blocks), and the crosstalk between the mTOR and cAMP-PKA pathways. Dr. Guan also solved the first-ever three-dimensional structure of a GTPase dimer, Gtr1/Gtr2, the yeast equivalent to human Rag proteins. Structure analysis resulting from this finding has led to further investigation by others in the field in the role of Rag proteins in cellular signaling.
More recently, Dr. Guan received a FY12 TSCRP Idea Development Award to build on the findings from his FY08 Award, described above, to study the molecular mechanism and biological functions of mTORC1 regulation by the cAMP-PKA pathway. Thus far, he has demonstrated that PKA is mediating the effect of cAMP on mTORC1 inhibition. His recent work has revealed that the amino acid glutamine activates mTORC1 in a Rag-independent mechanism in which Arf1 GTPase plays a key role.
With TSCRP funding, Dr. Guan is continuing to advance our knowledge of the role of TSC in the mTOR pathway by identifying and characterizing many of the key regulators in the pathway. This understanding may lead to new therapeutic targets that will help alleviate some of the manifestations of tuberous sclerosis.
Publications:
Yang Q, et al. 2006. Identification of Sin1 as an essential TORC2 component required for complex formation and kinase activity. Genes and Dev 20:2833-2847.
Lee CH, et al. 2007. Constitutive mTOR activation in TSC mutants sensitizes cells to energy starvation and genomic damage via p53. EMBO J 26(23):4812-4823.
Li Y, et al. 2007. Bnip3 mediates the hypoxia-induced inhibition on mTOR by interacting with Rheb. J Biol Chem 282:35803-35813.
Kim E, et al. 2009. RAG GTPases in nutrient mediated TOR signaling pathway. Cell Cycle 8:1014-1018.
Li L, et al. 2009. Amino acid signaling to TOR activation: Vam6 functioning as a Gtr1 GEF. Mol Cell 35:543-545.
Li L, et al. 2010. Regulation of mTORC1 by the Rab and ARF GTPases. J Bio Chem 285:19705-19709.
Kang YJ, et al. 2011. The TSC1 and TSC2 tumor suppressors are required for proper ER stress response and protect cells from ER stress-induced apoptosis. Cell Death Differ 18:133-144.
Kim J, et al. 2011. Amino acid signaling in mTOR activation. Ann Rev Biochem 80:1001-1032.
Gong R, et al. 2011. Crystal structure of Gtr1p-Gtr2p complex reveals new insights into the amino-acid induced TORC1 activation. Genes Dev 25:1668-1673.
Zheng M, et al. 2011. Inactivation of Rehb by PRAK-mediated phosphorylation is essential for energy-depletion-induced suppression of mTORC1. Nature Cell Biol 13:263-272.
Jewell JL, et al. 2013. Amino acid signaling upstream of mTOR. Nature Rev Mol Cell Biol 14:133-139.
Jewell JL, et al. 2013. Nutrient signaling to mTOR and cell growth. Trends Biochem Sci 38(5):233-242.
Jewell JL, et al. 2015. Differential regulation of mTORC1 by leucine and glutamine. Science 347(6218):194-198.
Links:
Public and Technical Abstracts: Regulation of TSC1/TSC2 Stability and Rheb GTP Level by Herc1
Public and Technical Abstracts: BIG1 as a Potential Guanine Nucleotide Exchange Factor for Rheb
Public and Technical Abstracts: Function of p53 in Regulation of TSC Mutant Cell Apoptosis
Public and Technical Abstracts: Regulation of the mTOR Pathway by a Novel Rheb Binding Protein BNIP3
Public and Technical Abstracts: Regulation of mTOR by Nutrients
Public and Technical Abstracts: Crosstalk between mTORC1 and cAMP Signaling














