Posted January 31, 2013
Kevin Ess, M.D., Vanderbilt University Medical Center
 Tuberous Sclerosis Complex (TSC) is a genetic disorder resulting from loss of function of the TSC1 or TSC2 genes and characterized by the development of nonmalignant tumors in many different organs, primarily in the brain where they are called "tubers", heart, kidney, skin, and lungs. While some pharmacological targets for the treatment of TSC have been identified, available therapies are largely ineffective in TSC patients with intractable epilepsy, autism, and developmental delays. Additional tools to facilitate the understanding of TSC gene function and to catalyze the discovery of novel therapeutic strategies are needed. Mouse models of TSC are limited because deletion of the Tsc1 or Tsc2 genes is lethal, and mice that are heterozygous for Tsc1 or Tsc2 mutations have a very mild disease phenotype. Dr. Kevin Ess at the Vanderbilt University Medical Center has developed a zebrafish model of TSC by inactivating the tsc2 gene. Zebrafish are an attractive research model because they produce a large number of embryos, are transparent, simplifying study of their internal development, and can be easily modified genetically, which aids in the study of gene interactions.
Tuberous Sclerosis Complex (TSC) is a genetic disorder resulting from loss of function of the TSC1 or TSC2 genes and characterized by the development of nonmalignant tumors in many different organs, primarily in the brain where they are called "tubers", heart, kidney, skin, and lungs. While some pharmacological targets for the treatment of TSC have been identified, available therapies are largely ineffective in TSC patients with intractable epilepsy, autism, and developmental delays. Additional tools to facilitate the understanding of TSC gene function and to catalyze the discovery of novel therapeutic strategies are needed. Mouse models of TSC are limited because deletion of the Tsc1 or Tsc2 genes is lethal, and mice that are heterozygous for Tsc1 or Tsc2 mutations have a very mild disease phenotype. Dr. Kevin Ess at the Vanderbilt University Medical Center has developed a zebrafish model of TSC by inactivating the tsc2 gene. Zebrafish are an attractive research model because they produce a large number of embryos, are transparent, simplifying study of their internal development, and can be easily modified genetically, which aids in the study of gene interactions.
Dr. Ess received an Idea Development Award from the TSC Research Program to characterize brain abnormalities, hamartoma formation, and gene interactions in the mTOR signaling pathway in tsc2-deficient zebrafish. He has found alterations in brain development in tsc2-deficient zebrafish, including increased cell size in the brain and poor organization of forebrain neurons, leading to extensive disorganization of gray and white matter and ectopically positioned cells. Tsc2-deficient fish also have increased mTOR Complex 1 (TORC1) activity, which was reversed by treatment with rapamycin or expression of zebrafish or human TSC2 mRNA. Further, transplantation of fluorescently labeled tsc2-deficient cells into wild-type zebrafish embryos resulted in wild-type neurons in the host brain to become mispositioned. Eight months following transplantation, tsc2-deficient cells were seen in multiple organs and displayed increased TORC1 signaling. One year after cell transplantation, apparent hamartomas of tsc2-deficient cells were seen in the brain. These findings lead Dr. Ess to conclude that the tsc2 mutant cells have cell autonomous and possibly non-cell autonomous mechanisms that lead to aberrant brain development. Additional experiments to better understand the role of tsc2 in zebrafish development are ongoing. The knowledge gained from these studies will not only impact the TSC community but will provide insight into the role of mTOR signaling in development and disease pathogenesis.
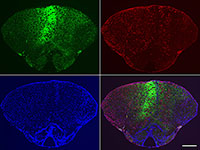
Brain of wild type zebrafish 8 months following the transplantation of tsc2 homozygous mutant cells labeled with green fluorescent protein (GFP). Upper left: GFP (green); upper right: phospho-S6, a marker of mTORC1 signaling (red); lower left: DAPI to identify cell nuclei (blue); lower right: merged image. Scale bar equals 200 μm.
Links:
Public and Technical Abstracts: Neural Development in tsc2-Deficient Zebrafish














