


Posted April 16, 2015
Duojia Pan, Ph.D., Johns Hopkins University
 Neurofibromatosis (NF) is a genetic disease that disturbs cell growth in the nervous system, leading to tumor growth on nerve tissues including the brain, spinal cord, and nerves, which can be benign or malignant. Depending on the location of these tumors, symptoms can range from hearing loss and learning impairment to severe disability, loss of vision, and severe pain. These clinical manifestations of neurofibromatosis vary among the three distinct types of NF, which include neurofibromatosis type I (NF1), neurofibromatosis type II (NF2), and schwannamatosis, and new therapeutics for each type of NF are greatly needed. Although much is known about the genetic pathways regulated by the neurofibromin 1 gene, which is the genetic cause of NF1, much less is known about NF2. NF2 is caused by mutations in the Merlin gene, although the function and genetic signaling resulting from these mutations are largely unknown, hindering the development of effective therapeutics for NF2.
Neurofibromatosis (NF) is a genetic disease that disturbs cell growth in the nervous system, leading to tumor growth on nerve tissues including the brain, spinal cord, and nerves, which can be benign or malignant. Depending on the location of these tumors, symptoms can range from hearing loss and learning impairment to severe disability, loss of vision, and severe pain. These clinical manifestations of neurofibromatosis vary among the three distinct types of NF, which include neurofibromatosis type I (NF1), neurofibromatosis type II (NF2), and schwannamatosis, and new therapeutics for each type of NF are greatly needed. Although much is known about the genetic pathways regulated by the neurofibromin 1 gene, which is the genetic cause of NF1, much less is known about NF2. NF2 is caused by mutations in the Merlin gene, although the function and genetic signaling resulting from these mutations are largely unknown, hindering the development of effective therapeutics for NF2.
With support from a CDMRP FY09 Neurofibromatosis Research Program (NFRP) Investigator-Initiated Research Award, Dr. Duojia Pan and his research team at Johns Hopkins University sought to use Drosophila to identify downstream genetic pathways controlled by Merlin. Drosophila, commonly known as the fruit fly, has many evolutionarily conserved genes, including Merlin, that are strikingly similar to those in humans. Through this work, Pan and his team revealed multiple layers of evidence linking Merlin to the Hippo genetic signaling pathway, an important signaling pathway in cancer development. The Hippo pathway involves a signaling cascade of genes including Hippo (Hpo), Warts (Wts), and YAP. First, they discovered that YAP, which is elevated in many human cancers, genetically interacts with Merlin and the two genes function antagonistically to regulate mammalian tissue growth. Second, they showed that a protein complex, which consists of Kibra, Merlin, and Expanded, regulates Hippo signaling by targeting the Wts protein, and these proteins cooperate with each other to suppress tumor formation.
Dr. Pan currently has an FY13 NFRP Investigator-Initiated Research Award to continue these studies. Research supported by this award will use the molecular data identified in his Drosophila studies to develop targeted therapies against NF2. Dr. Pan plans on testing an inhibitor of YAP that was identified in his laboratory as well as identifying additional targets and potential inhibitors against those targets. These are the first studies to test drugs that specifically target the Merlin gene pathway and may lead to much-needed therapeutics for the treatment of NF2.
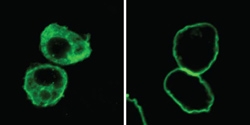
Merlin is a Matchmaker, not a Magician
The protein "arranges" other protein interactions to control growth and prevent cancer. Without Merlin around, Warts (green) is located mostly in the watery interior of the cell (left). When Merlin is there, Warts is localized to the outer envelope of the cell where Merlin resides (right). Credit: Duojia Pan, courtesy of Cell Press.
Publications:
Yin F, et al. 2013. Spatial organizational of hippo signaling at the plasma membrane mediated by the tumor suppressor merlin/NF2. Cell 154:1342-1355.
Zhang N, et al. 2010. The merlin/NF2 tumor suppressor functions through the YAP oncoprotein to regulate tissue homeostasis in mammals. Dev Cell 19:27-38.
Pan D. 2010. The hippo signaling pathway in development and cancer. Dev Cell 19:491-505.
Links:
Public and Technical Abstracts: Developing Molecular Targeted Therapies against NF2














