


Posted June 3, 2014
Lu Le, M.D., Ph.D., University of Texas Southwestern Medical Center at Dallas
 Neurofibromatosis type 1 (NF1) is one of the most common genetic disorders, affecting approximately 1 in 3,500 people worldwide. NF1 is caused by a mutation - that can be hereditary or spontaneous - in the neurofibromin 1 gene and often results in dermal or plexiform neurofibromas. Although neurofibromas are benign tumors, they can cause physical disfigurement and impairment of organ or neural function, and the plexiform neurofibromas have a 10% risk of developing into life-threatening malignant peripheral nerve sheath tumors (MPNSTs), which is the main cause of mortality for NF1 patients. The underlying mechanisms mediating initiation and progression of these tumors are largely unknown, and this knowledge is crucial for the development of new therapeutics for individuals with NF1. Dr. Lu Le, of the University of Texas Southwestern Medical Center, has made great strides in the NF1 field by determining the cell of origin for these complex tumors to study their biology.
Neurofibromatosis type 1 (NF1) is one of the most common genetic disorders, affecting approximately 1 in 3,500 people worldwide. NF1 is caused by a mutation - that can be hereditary or spontaneous - in the neurofibromin 1 gene and often results in dermal or plexiform neurofibromas. Although neurofibromas are benign tumors, they can cause physical disfigurement and impairment of organ or neural function, and the plexiform neurofibromas have a 10% risk of developing into life-threatening malignant peripheral nerve sheath tumors (MPNSTs), which is the main cause of mortality for NF1 patients. The underlying mechanisms mediating initiation and progression of these tumors are largely unknown, and this knowledge is crucial for the development of new therapeutics for individuals with NF1. Dr. Lu Le, of the University of Texas Southwestern Medical Center, has made great strides in the NF1 field by determining the cell of origin for these complex tumors to study their biology.
With funding from an FY12 NFRP New Investigator Award, Dr. Le and colleagues sought to determine where neurofibromas come from and his research group identified a population of neural crest-related stem cells in the skin, called skin-derived precursors (SKPs), as the cell of origin for the dermal neurofibroma. His next aim is to study how these SKPs interact with other cells in their microenvironment with the goal of identifying therapeutic interventions to target these cell interactions. To do this, he developed a novel 3D cell culture model of neurofibroma using SKPs and the cells present in their microenvironment. For the first time, this model allowed for all stages of neurofibroma evolutionary development, from tumor initiation to tumor progression, to be observed and studied in tissue culture dishes as well as for transplantation into animals for in vivo analysis. In the course of these SKP and neurofibroma studies, the investigators accidentally found that they could readily generate MPNSTs from SKPs deficient in both neurofibromin and P53 genes. Dr. Le took advantage of this novel MPNST mouse model that permits the study of tumor evolution to perform transcriptome analysis, a method that examines changes in all forms of RNA levels, and identified that BRD4, a BET (bromodomain and extraterminal) family member, was highly upregulated in MPNSTs. Additionally, pharmacologic inhibition of BRD4 with compound JQ1 significantly inhibited the tumorigenesis of NF1-associated MPNSTs in this mouse model, providing strong evidence for evaluating the use of BET bromodomain inhibitors for NF1 patients who develop MPNSTs.
In collaboration with Dr. Luis Parada's lab, using the same model system, the team also found that a G protein-coupled receptor, CXCR4, is also highly expressed in MPNSTs compared to nonmalignant neurofibromas. Furthermore, they also found that a CXCR4 pharmacologic inhibitor, AMD3100, effectively inhibited tumor growth in vitro and in vivo, pointing to another potential new therapeutic target for MPNSTs.
Through his NFRP-sponsored research, Dr. Le and colleagues have discovered two novel therapeutic targets, CXCR4 and BRD4, for NF1-associated MPNSTs. Moreover, the 3D cell culture and NF1 animal models that Dr. Le has established allow for increased biologic understanding of neurofibroma evolution, from tumor initiation to malignant transformation, and could result in the discovery of many more therapeutic targets and future testing for drugs to treat NF1.
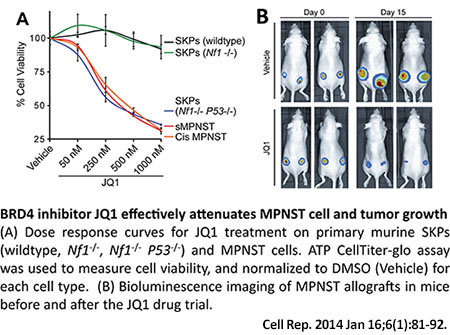
Publications:
Patel AJ, Liao CP, Chen Z, Liu C, Wang Y, and Le LQ. 2014. BET bromodomain inhibition triggers apoptosis of NF1-associated malignant peripheral nerve sheath tumors through Bim induction. Cell Reports 6(1):81-92.
Mo W, Chen J, Patel A, Zhang L, Chau V, Li Y, Cho W, Lim K, McKay R, Lev D, Le LQ and Parada L. 2013. CXCR4/CXCL12 mediate autocrine cell cycle progression in NF1-associated malignant peripheral nerve sheath tumors. Cell 152(5):1077-1090.
Chen Z, Pradhan S, Liu C, and Le LQ. 2012. Skin-derived precursors as a source of progenitors for cutaneous nerve regeneration. Stem Cells 30(10):2261-2270.
Links:














