


Posted February 24, 2015
Sheila Nirenberg, Ph.D., Neuroscientist, Cornell University
 There are few treatment options for advanced stage blindness due to combat-based eye injuries or retinal degenerative diseases, largely due to extensive tissue loss and, more specifically, loss of photoreceptors. Prosthetic devices have the ability to bypass the damaged tissue and directly stimulate the retina's surviving cells, but currently available devices are minimally effective, with low resolution and limited signal processing. Dr. Sheila Nirenberg of Cornell University received a Fiscal Year 2011 Vision Research Program Translational Research Award to develop a new kind of prosthesis that has the potential to be much more effective than standard vision restoration approaches.
There are few treatment options for advanced stage blindness due to combat-based eye injuries or retinal degenerative diseases, largely due to extensive tissue loss and, more specifically, loss of photoreceptors. Prosthetic devices have the ability to bypass the damaged tissue and directly stimulate the retina's surviving cells, but currently available devices are minimally effective, with low resolution and limited signal processing. Dr. Sheila Nirenberg of Cornell University received a Fiscal Year 2011 Vision Research Program Translational Research Award to develop a new kind of prosthesis that has the potential to be much more effective than standard vision restoration approaches.
There are two main components of a vision prosthetic device: the signal processing component, which converts images into signals, and the transducer, which simulates the retina output cells (i.e., ganglion and bipolar cells) to relay those signals to the brain. Early prosthetic devices utilized a camera to collect images and convert them into electrical signals, which would then feed into electrodes to stimulate the cells; at best, however, vision included the ability to see bright lights and high contrast edges. Then optogenetics arrived on the scene a decade ago, introducing a new strategy for controlled stimulation in which a gene with light sensitive properties would be inserted into retinal cells, thereby enabling the cells to be stimulated by varying degrees of light. While this method of stimulation improved resolution, the images themselves were not natural (i.e., signals from the retina to the brain were not "normal").
Dr. Nirenberg realized that in addition to improving resolution, it was also necessary to accurately process the signals. She had previously developed an algorithm in a mouse model to convert visual input (i.e., images) into a neural code that can be understood by the brain. Using this neural code as an "encoder" during the signal processing step, in combination with optogenetics (inserting the light sensitive protein, channelrhodopsin [ChR] into retinal cells), she demonstrated that her prosthetic device could produce image representation much closer to normal vision.
Dr. Nirenberg is currently adapting and applying her discoveries in neural coding so that they can be used in humans. In an important step toward this goal, Dr. Nirenberg has adapted her prosthetic system to another animal model that shares greater similarity with the human visual system. First, she has identified the retinal code for this model, so that the images can be correctly interpreted by the brain. She has also identified a vector capable of expressing ChR in ganglion cells in sufficient quantities to stimulate the retina into relaying the code to the brain. A prototype device was constructed using the neural code that is capable of stimulating more than 1000 cells in the central retina in real time. Results demonstrate that the device produced normal firing patterns (i.e., signals) in these animals. Further modifications are being made to adapt this prosthetic system for clinical use. Dr. Nirenberg has teamed with gene therapy expert Dr. Sam Jacobson of the Scheie Eye Institute at the University of Pennsylvania to conduct a Phase I/II clinical trial, which is anticipated to begin in 2016 or 2017.
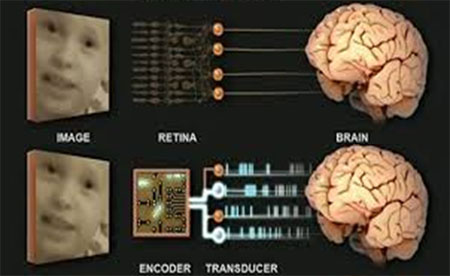
Top, in retinal degenerative diseases, suffered by civilians and veterans alike, the photoreceptors and neighboring cells degenerate, but the retina's output cells remain largely intact. This opens the door to a prosthetic treatment. A device can jump over the damaged tissue and provide direct stimulation to the output cells, allowing them to send signals to the brain. Bottom, the device mimics the operations performed by the retina, that is, it transforms images into the code used by the output cells, and then it stimulates the output cells so they can send the code onto the brain. As shown, the device has two parts: a) a camera/encoder component, which takes images in and converts them into the code, and b) a transducer, which drives the output cells to send the code onward.














