Traumatic Brain Injury and Psychological Health
Clinical Trial of Sildenafil Citrate for Treatment of Cerebrovascular Dysfunction in Chronic Traumatic Brain Injury



Posted April 5, 2023
Ramon Diaz-Arrastia, M.D., Ph.D., University of Pennsylvania
Kimbra Kenney, M.D., Walter Reed National Military Medical Center
Priyanka Kalyani, CCRC-PM, B.S., University of Pennsylvania

(Photo Provided)
Globally, traumatic brain injury (TBI) affects over 70 million people each year. TBI results from trauma to the head such as falls, motor vehicle accidents, gunshot wounds, etc., that affect brain function and may cause a variety of long-term cognitive (memory and thinking), behavioral, mood, and sensory symptoms. These may include loss of consciousness, nausea, vomiting, amnesia, aggression, and other problems that can range from mild to severely debilitating.
Despite the high numbers and frequent occurrence of TBI, the cellular mechanisms underlying TBI-related brain damage are poorly understood. More importantly, there are currently no approved drug therapies to directly treat TBI. This is due, in part, to the complexity of any brain injury and different brain components (neurons, axons, blood vessels) variably impacted, but also because we do not yet have good non-invasive ways of determining and measuring the severity of injury in each of these brain components. There is an urgent need for specific biomarkers (substances that can be measured independently of the injury) to identify subgroups of TBI patients based on the severity of injury in each brain component that will best respond to targeted therapies.
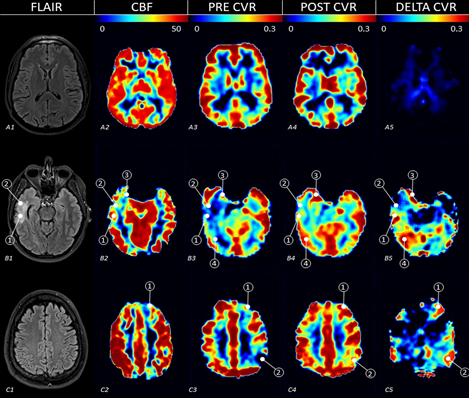
In previous studies, Dr. Diaz-Arrastia and his team identified cerebrovascular reactivity (CVR), a measure of vascular health in the brain, as a unique biomarker to test candidate interventions for traumatic cerebrovascular injury (TCVI). TCVI is damage to blood vessels (the vascular component) in the brain due to head trauma and has been associated with higher rates of TBI-related disability and death. The group used magnetic resonance imaging (MRI) to compare CVR measurements between TBI patients and healthy cohorts. They determined that CVR and blood flow are decreased in chronic TBI patients. The team conducted a Phase 2a clinical trial (clinincaltrials.gov, NCT01762475) to test the effect of a U.S. Food and Drug Administration-approved drug, sildenafil (Viagra®/Revatio®), to improve blood flow in the brain following a TBI. Sildenafil works by blocking the action of an enzyme in the smooth muscle of the smallest brain vasculature, which then allows the blood vessels to dilate and increase blood flow. Upon completion of the clinical trial, the researchers found that a single treatment with sildenafil increased CVR in TBI patients (Figure 1). They also tested patients' tolerance to a low dose of twice-daily sildenafil for 8 weeks to ensure it was safe in TBI patients. The treatment was well-tolerated, and participants reported improved chronic TBI symptoms.
With funding from a fiscal year 2021 Clinical Trial Award from the TBI and Psychological Health Research Program (TBIPHRP), Dr. Diaz-Arrastia and his team are building on that previous work in a Phase 2b clinical trial (clinicaltrails.gov registry number pending). In a large number of TBI patients, they will assess the safety and tolerability of three sildenafil doses (that are currently used in adults with pulmonary hypertension). The goal is to identify the lowest effective dose to maximally improve or normalize blood flow caused by TCVI and then measure how that impacts long-term TBI symptoms after 4 weeks of daily sildenafil. The team is currently enrolling participants with chronic TBI (https://clinicalresearch.itmat.upenn.edu/clinicaltrial/7317/medical-research-phase-2-randomized-controlled/). If the current clinical trial proves successful, sildenafil may represent a powerful treatment opportunity for TBI among Veterans, active-duty Service Members, and civilians.
References:
Kenney K, Amyot F, Haber M, et al. 2015. Cerebral Vascular Injury in Traumatic Brain Injury. Experimental Neurology. 275(3):353-366.
Amyot F, Kenney K, Moore C, et al. 2018. Imaging of Cerebrovascular Function in Chronic Traumatic Brain Injury. Journal of Neurotrauma. 35:1116-1123.
Kenney K, Amyot F, Moore C, et al. 2018. Phosphodiesterase-5 Inhibition Potentiates Cerebrovascular Reactivity in Chronic Traumatic Brain Injury. Annals of Clinical and Translational Neurology. 5(4):418-428.
https://clinicaltrials.gov/ct2/show/NCT01762475?term=NCT01762475&draw=2&rank=1
Last updated Friday, September 19, 2025
 An official website of the United States government
An official website of the United States government
 ) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
