Tick-Borne Disease
Wearable Devices for Controlled Release of Tick Repellents



Posted October 19, 2022
Noel Elman, Ph.D., GearJump Technologies, LLC

(Photo provided)
As tick populations continue to grow and expand geographically, tick-borne diseases (TBDs), including Lyme disease, represent a significant risk for U.S. military personnel operating in these environments. According to the Armed Forces Health Surveillance Branch (AFHSB), approximately 6,0001 Service Members and 56,0001 of their beneficiaries were diagnosed with a TBD between 2006 and 2020, with Lyme disease representing approximately 80%1 of those diagnoses. Lyme disease accounted for about 44%2 of all reportable medical event cases of vector-borne diseases (confirmed, probable, and suspected) among active and reserve components between 2016 and 2020. Lyme disease is also the most common vector-borne disease in the U.S., with recent estimates suggesting approximately 476,000 Americans are diagnosed and treated for Lyme disease each year.3

Figure 1. Adaptive Barrier-Controlled Release Device (AB-CRD). Actual prototype of an AB-CRD attached to a standard issue Army boot. The device is easily integrated with Soldier's boots to protect against ticks during training and deployment. (Photo provided)
The health and financial costs of TBDs for civilians and military personnel can be overwhelming. Symptoms of Lyme disease may include a rash with a characteristic "bullseye” appearance, fever, swollen lymph nodes, chills, dizziness, shortness of breath, severe headaches and neck stiffness, fatigue, heart palpitations/irregular heartbeat, arthritis, neuroinflammation, and pain in muscles, tendons, bones, joints, and nerves.4 If Lyme disease is diagnosed early and treated with antibiotics, patient outcomes are generally positive. However, with improper or lack of treatment, some patients can develop post-treatment Lyme disease syndrome (PTLDS) with persistent symptoms, such as pain, fatigue, and/or difficulty thinking.5 Infected ticks can also transmit other bacterial, viral, and parasitic pathogens, resulting in various diseases including Rocky Mountain Spotted Fever, ehrlichiosis, babesiosis, anaplasmosis, Powassan encephalitis, and others.6 While current methods of tick-bite prevention include sprays, topical creams, and permethrin-treated clothing, continued application is required for long-term effectiveness. Dr. Noel Elman and the team at GearJump Technologies, LLC, have been working to develop a long-term, efficient, and safe solution to repel ticks and prevent transmission of tick-borne pathogens by creating the next generation of personal protective gear, dubbed Adaptive Barrier-Controlled Release Devices (AB-CRDs).
With support from a Fiscal Year 2018 Investigator-Initiated Research Award from the Congressionally Directed Medical Research Programs' Tick-Borne Disease Research Program (TBDRP), Dr. Elman has made substantial progress in designing Micro-Electro-Mechanical-Systems (MEMS) as key technological components to provide controlled release of repellants in AB-CRDs for personal protection against ticks. The MEMS-based devices provide sustained and smart delivery of spatial repellents (to create a barrier around the device and wearer) and contact repellents (to deter ticks from biting the wearer). The MEMS devices are integrated with electronics for programming, sensor integration, remote activation, and autonomous operation and can be embedded in straps or bands, making them easy to wear outdoors (Figure 1). They can be used for 1 to 3 weeks with a single-use cartridge containing multiple reservoirs and are either automatically or remotely triggered to release tick repellent. Dr. Elman compares the function of AB-CRDs to "drip irrigation” where a slow, steady level of repellent expulsion creates a safe, cost-effective, and sustainable solution to tick prevention.
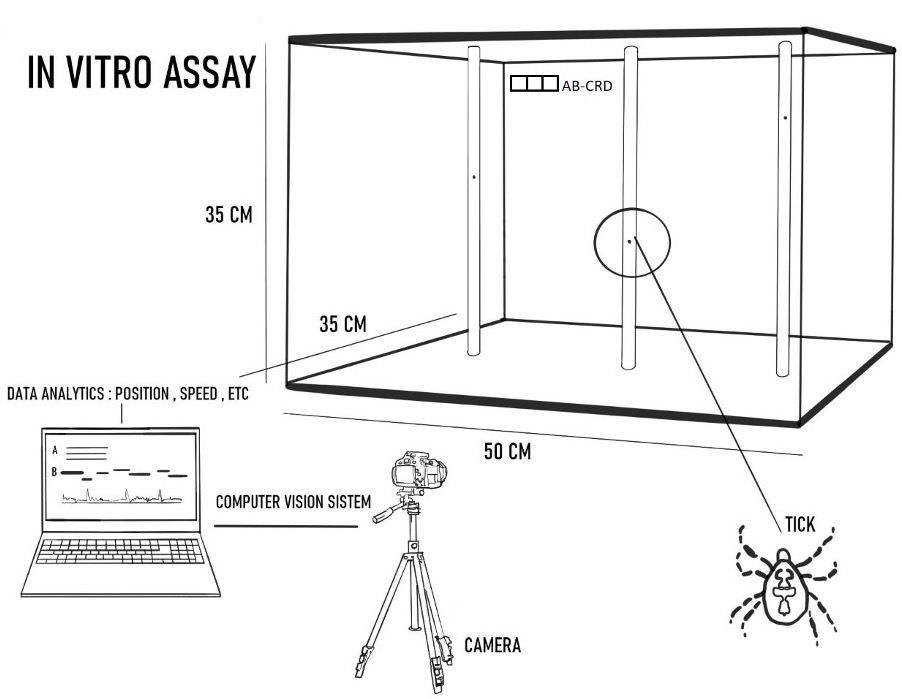
Figure 2. Experimental Setup Employing the Vertical Tick Assay for Evaluation of Spatial Repellents (VTA-ESR). Several active ingredients (AIs) were tested as tick repellants in the AB-CRDs to determine their effects on tick host-seeking behavior. (Graphic provided)
Dr. Elman is collaborating with experienced research partners from the U.S. Department of Agriculture (USDA) and the University of Massachusetts Amherst to determine the most effective combination of tick repellent to employ in the devices. Considerations include finding a tick repellent with broad applicability against several species of ticks, overall safety, and efficacy of preventing tick dispersal and attachment. The GearJump Technologies team and its research partners investigated the effects of using various active ingredients (AIs) in the AB-CRDs, including transfluthrin, metofluthrin, nootkatone, and DEET. The AIs were tested against adult females of three common species of ticks (American dog tick, Lone star tick, black-legged tick). The novel Vertical Tick Assay for Evaluation of Spatial Repellents (VTA-ESR) was utilized to provide a quantitative model of innate host-seeking tick behavior, repellency, and detachment (Figure 2). Results suggest that AB-CRDs cause changes in tick movement velocity, displacement, detachment, and rate of successful vertical climbing. Future studies will consider binding properties of AIs to clothing, hair, and skin, different concentrations and combinations of AIs, as well as release rates, and environmental factors such as abrasion, temperature, wind, and humidity. Dr. Elman's team has also partnered with the U.S. Army Combat Capabilities Development Command Soldier Center in Natick, Massachusetts, to obtain feedback on form, fit, function, and end user priorities/needs of the CRDs. Soldiers have expressed growing concern for tick populations and tick bites while providing valuable insight about durability, environmental concerns, and the use of multiple devices in enclosed spaces. These factors will be considered when moving the device design forward.
Overall, the potential impact of AB-CRDs is significant, as they would protect Service Members from tick bites, reducing the incidence of TBDs in the military. Most recently, GearJump Technologies has received follow-on funding from the Deployed Warfighter Protection Program administered by the Armed Forces Pest Management Board to aid in further development and validation of similar wearable devices. While additional studies are necessary to optimize the devices, Dr. Elman hopes they can be used by civilians and Service Members to prevent pathogen transmission and reduce the negative impacts of Lyme disease and other TBDs in areas with pervasive tick populations.
References:
1Data from the Armed Forces Health Surveillance Branch (AFHSB) for the years 2006-2020
2AFHSB's February 2021 Medical Surveillance Monthly Report
4Signs and Symptoms of Untreated Lyme Disease
5Post-Treatment Lyme Disease Syndrome
Links:
Last updated Wednesday, January 7, 2026
 An official website of the United States government
An official website of the United States government
 ) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
