Tick-Borne Disease
Scientist Working to Improve Diagnosis of Lyme Disease and Other Tick-Borne Illnesses



Posted December 3, 2019
Charles Chiu M.D., Ph.D., University of California, San Francisco

Documented cases of tick-borne diseases (TBDs), such as spotted fever rickettsiosis, anaplasmosis, ehrlichiosis, babesiosis, tularemia, Powassan virus disease, and Lyme disease are on the rise, with Lyme disease being the most prevalent. In 2017, the Centers for Disease Control and Prevention reported a total of 59,349 cases of TBDs, representing an increase of just over 10,000 cases from the previous year.1 As tick populations continue to expand and case numbers continue to rise, the need for sensitive, specific, and rapid diagnostics assays becomes more urgent as many TBD cases go unreported, undiagnosed, or misdiagnosed.
The currently available clinical test for the diagnosis of Lyme disease is a two-tier serology-based screening that relies solely on the patient’s antibody response. The first tier is an enzyme linked immunosorbent assay (ELISA) (or immunofluorescence assay) to evaluate the patient’s blood for the presence of antibodies that are specific for the bacterium that causes Lyme disease (Borrelia burgdorferi) If positive or ambiguous, the patient’s samples are then sent for a second tier Western blot (or possibly the recently Food and Drug Administration-approved Tier 2 ELISA) in which a more specific antibody assay is performed to evaluate the presence of immunoglobulin (Ig) M or IgG. Unfortunately, the sensitivity of this two-tier system is relatively low during early infection, ranging from 30-40%, as the patient is only just mounting an antibody response.2 Notably, treatment in the early phase of infection prevents potential late-stage complications of disseminated Lyme disease, including arthritis, neurological symptoms, and cardiac arrhythmias.3 When patients present later, the two-tier testing has 70-100% sensitivity2; however, diagnostics solely based on serology may be unable to distinguish between current (active), recent, or past infection. For example, Borrelia antibodies that persist from a past infection may lead to a false positive result.2 The lack of accurate tests to diagnose early infection can result in delayed or inappropriate treatment that may put the patient at risk for more severe outcomes, including Lyme-associated arthritis, carditis, neurologic complications (such as neuroborreliosis), and possibly post-treatment Lyme disease syndrome, commonly referred to as “chronic Lyme,” for which no specific, approved therapeutic regimen exists. Improved diagnostic assays would increase clinician confidence in initiating appropriate therapy, thus reducing the incidence of adverse outcomes in patients with treatable infections.
Charles Chiu M.D., Ph.D., Professor of Laboratory Medicine and Medicine/Infectious Diseases, Director of the University of California, San Francisco (UCSF) - Abbott Viral Diagnostics and Discovery Center and Associate Director of the UCSF Clinical Microbiology Laboratory at the University of California, San Francisco, was the recipient of a Fiscal Year 2016 Tick-Borne Disease Research Program (TBDRP) award. Dr. Chiu and his team aim to develop assays for the diagnosis of early Lyme disease and other tick-borne infections and the differentiation of Lyme disease from other illnesses with similar clinical presentation. While they are employing multiple approaches to address these gaps in the field of TBDs, Dr. Chiu’s TBDRP-funded award has largely supported efforts concentrated on (1) the identification of a human host molecular biomarker signature that is distinct for Lyme disease, (2) the use of metagenomic sequencing with spiked primer enhancement (MSSPE) to enrich for TBDs from patient samples, and (3) the evaluation of a novel “TickChip” using clustered regulatory interspaced short palindromic repeats (CRISPR)-associated (Cas) gene technology and developed under industrial partnership that would facilitate the simultaneous diagnosis of almost all TBDs.
Toward biomarker identification, Dr. Chiu and his team have generated transcriptomic profiles of whole blood and human peripheral blood mononuclear cells from patients with confirmed Lyme disease, sepsis, influenza, or Babesia infection (the second most common tick-borne disease), as well as age, sex, and geography-matched blood donors. Using machine learning algorithms, a predictive model was constructed that can discriminate between Lyme disease, sepsis, influenza, and controls with 87% accuracy (Figure 1).
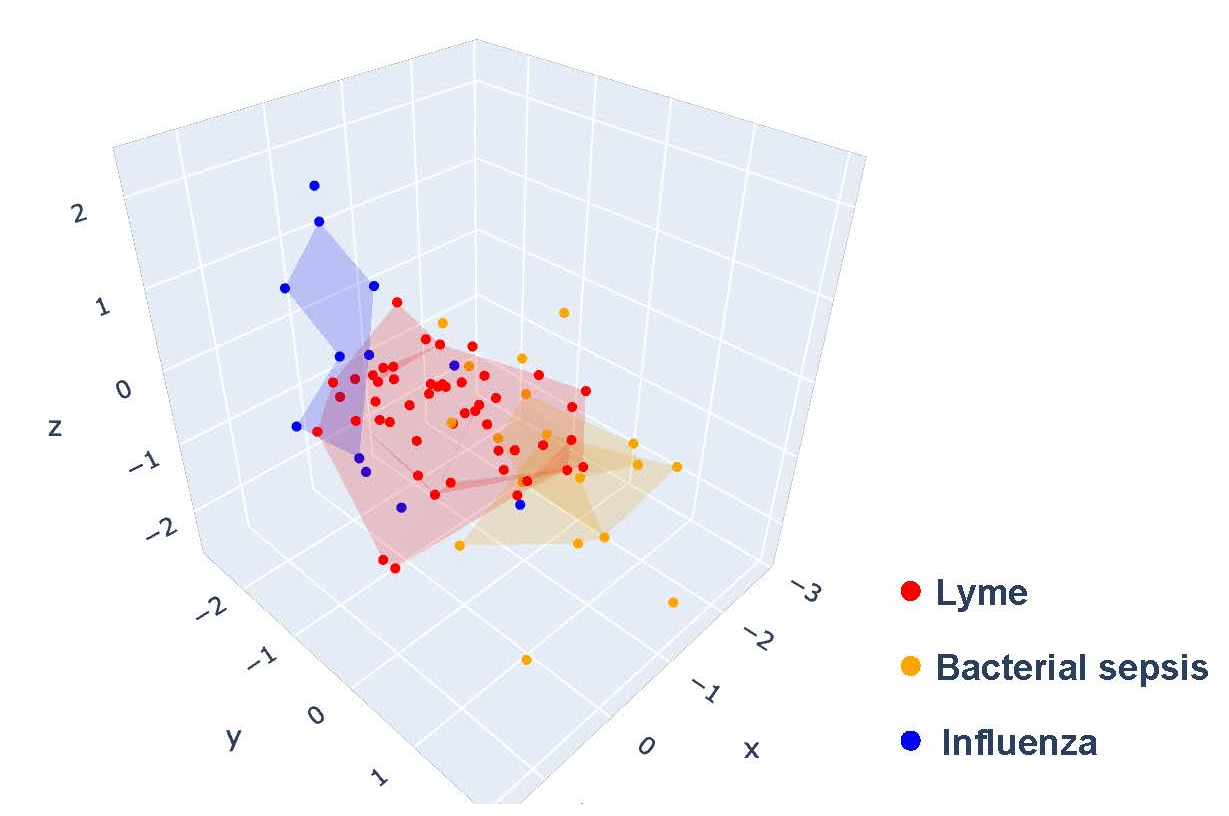
Other study aims involve metagenomics sequence analysis and enrichment for defined panels of pathogens. Dr. Chiu’s laboratory has recently developed a metagenomic sequencing with spiked primer enrichment (MSSPE) technique that includes “spiked primers” specific for targeted viral pathogens.4 Dr. Chiu and his team were able to use MSSPE flaviviral spiked primers to successfully detect the presence of tick-borne infection from Powassan virus and recovered more of the viral genome as compared to random metagenomics sequencing alone (Figure 2). There are vast implications for the MSSPE assay, as Dr. Chiu has successfully used this technology for the enrichment and detection of other emerging viruses such as Ebola and Zika virus and hopes to bring this technology to the forefront of clinical diagnostics. In addition, in collaboration with Mammoth Biosciences, Inc., Dr. Chiu is working on a “TickChip” that would allow for the simultaneous detection of nearly all tick-borne pathogens from whole blood or other clinical samples. Using CRISPR-Cas technology, this diagnostic assay allows for rapid and specific enrichment, amplification, and detection of bacterial, parasitic, and viral tick-borne pathogens. Using this technology, Dr. Chiu has demonstrated that as few as 100 genome copies of Borrelia burgdorferi could be specifically detected and could be distinguished from its closely related pathogenic neighbor Borrelia miyamotoi, despite having only one nucleotide mismatch in the target gene region.
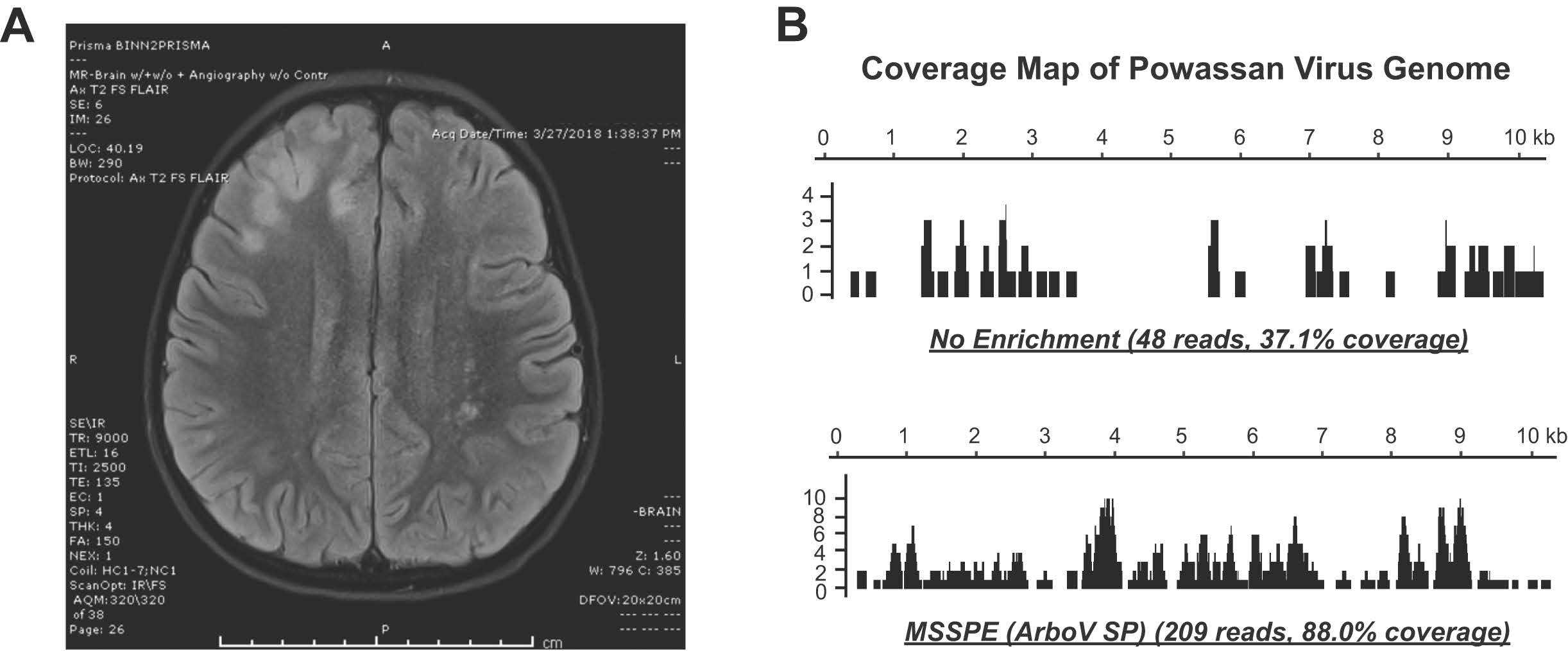
Figure 2: Metagenomic diagnosis of tick-borne Powassan virus infection. A 5-year-old boy presented with fever, vomiting, and seizures. Magnetic resonance imaging of the brain (A) showed focal lesions in the R frontal lobe. Metagenomic sequencing of cerebrospinal fluid (CSF) was use to diagnose Powassan virus infection. Enrichment of the viral genome using flaviviral spiked primers (B) enabled accurate genotyping of this tick-borne viral pathogen.
Metagenomic next-generation sequencing for diagnosis of neurological infections such as Powassan virus has already been put into clinical practice at UCSF, and similar tests from blood (plasma) are expected to be launched in early 2020. Thus, the potential impact of these assays on the diagnosis of TBDs such as Lyme disease, would be tremendous and would benefit patients, clinicians, and researchers alike.
REFERENCES:
1 Centers for Disease Control and Prevention - Media Release; Record Number of Tickborne Diseases Reported in U.S. in 2017. https://www.cdc.gov/media/releases/2018/s1114-record-number-tickborne-diseases.html (published November 2018; accessed September 10, 2019).
2 Moore A, Nelson C, Molins C, et al. 2016. Current guidelines, common clinical pitfalls, and future directions for laboratory diagnosis of Lyme disease, United States. Emerg Infect Dis. Jul;22(7):1169–1177. doi: 10.3201/eid2207.151694. PMID: 27314832; PMCID: PMC4918152.
3 Clark RP and Hu LT. 2008. Prevention of Lyme disease and other tick-borne infections. Infect Dis Clin North Am. Sep;22(3):381-396, vii. doi: 10.1016/j.idc.2008.03.007. PMID: 18755380; PMCID: PMC3195518.
4 Deng X, Achari A, Federman S, et al. (2019). Metagenomic sequencing with spiked primer enrichment (MSSPE) for viral diagnostics and surveillance. Nature Microbiology (in press).
LINKS:
Dr. Chiu’s Laboratory Website: https://chiulab.ucsf.edu/
Last updated Thursday, December 5, 2024














