


Posted April 17, 2014
Radi Masri, University of Maryland, Baltimore
 1. What led you to this research question?
1. What led you to this research question?
Spinal cord injury (SCI) is associated with devastating consequences, one of which is the development of severe, intractable, debilitating chronic pain. Controlling this pain is a major clinical challenge, and available medications are rarely effective. One treatment, the electrical stimulation of the motor cortex, an area in the brain responsible for controlling movement, has shown great potential in reducing pain associated with SCI. However, results from clinical studies on motor cortex stimulation report mixed outcomes, and the applicability of this promising treatment is hampered by the lack of understanding of how stimulating the motor cortex ameliorates chronic pain. This lack of knowledge led us to investigate brain circuits involved in pain relief produced after electrical stimulation of the motor cortex.
2. What is the most important thing(s) that SCI researchers should know about the results of your SCIRP-funded research?
The results of this research project revealed a novel brain circuit that can be manipulated to manage pain following SCI (see Figure below). At the center of this circuit is an inhibitory nucleus in the brain called zona incerta. We have shown previously that the activation of zona incerta reduces activity in the thalamus and cortex, which are brain areas responsible for the processing of painful information. The zona incerta is directly connected to the motor cortex. We discovered that stimulating the motor cortex reduces pain by activating zona incerta neurons. Pain relief is dependent upon stimulus frequency and duration and can last for hours after the end of stimulation.
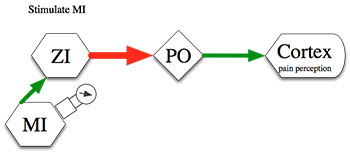
Figure: In normal conditions, pain information processed in the thalamus (PO) is regulated by powerful inhibition from zona incerta (ZI). In SCI pain, ZI activity is suppressed and therefore pain is enhanced. Electrical stimulation of the motor cortex (MI) enhances activity of zona incerta and restores inhibition in the thalamus. This will reduce the flow of painful information to the cortex and reduce pain perception. [Red represents inhibitory inputs; green: Excitatory Inputs; line thickness represents connection strength].
3. What is the most important thing(s) that individuals with SCI should know about the results of this research project?
Stimulating the motor cortex using small imperceptible electrical currents is a promising treatment modality that can be effective in controlling refractory SCI pain. This treatment can be administered non-invasively, with minimal side effects using direct current stimulation whereby electrical current is delivered through electrodes placed on top of the scalp, without implantation, to stimulate the motor cortex. Magnetic pulses can also be used to stimulate the motor cortex through the skull. This treatment, transcranial magnetic stimulation, is also non-invasive and associated with minimal side effects.
4. How is your research accelerating progress toward the SCIRP's vision to advance the treatment and management of SCI and ameliorate its consequences (including the potential short-term and long-term impact)?
The results of this project are a step forward in SCIRP's efforts to advance treatment and management of SCI pain. In the short term, we validate a potentially effective treatment for this debilitating condition and identify a novel brain circuit that is involved in the production of pain relief. This could lead, in the long term, to the development of new and innovative treatments that can be designed to target the circuit we describe to produce long lasting pain relief.
5. What has been the response to your research results from the SCI clinical community? Has there been movement toward increased use of motor cortex stimulation therapy for treatment of chronic neuropathic pain?
The results of this study have sparked great interest in our local clinical community and received significant interest when presented in national and international meetings. Several clinical studies are now being conducted, including one at the University of Maryland and one at Johns Hopkins University, to further evaluate the efficacy and to finesse clinical protocols for the use of this technique. Clinicians are starting to accept motor cortex stimulation as a promising modality for controlling treatment-resistant chronic pain.
6. What are the next steps to bringing your research closer to helping individuals with SCI? What are the next steps to transition motor cortex stimulation into clinical practice for the treatment of SCI-related neuropathic pain and what, if any, barriers are impeding this progress?
Dissemination of information and educating clinicians about the clinical efficacy and validity of this technique is a first step that will allow us to fully implement this treatment modality in SCI clinics. Making non-invasive stimulating devices available and training clinicians to use them effectively is another important step. Now that there is a clear mechanism illustrating how motor cortex stimulation can ameliorate pain, clinicians should be less reluctant to prescribe this modality of treatment for patients with SCI.
7. Is there any other information that you think readers should know about your work?
Pain reduction after motor cortex stimulation is transient and lasts for 30 to 90 minutes after the end of stimulation. Therefore, repeated, daily sessions of stimulation are necessary to control the pain. We are currently refining our motor cortex stimulation paradigms to produce prolonged pain relief with a minimal number of stimulation sessions.
Publications:
Cha M, Ji Y, and Masri R. 2013. Analgesic mechanisms of motor cortex stimulation in an animal model of spinal cord injury pain. J Pain 14:260-269.
Whitt J, Masri R, Pulimood N, et al. 2013. Pathological activity in mediodorsal thalamus of rats with spinal cord injury pain. J Neurosci 33:3915-3926.
Lucas JM, Ji Y, and Masri R. 2011. Motor cortex stimulation reduces hyperalgesia in an animal model of central pain. Pain 152:1398-1407.
Seminowicz DA, Jiang L, Ji Y, et al. 2011. Thalamocortical asynchrony in conditions of spinal cord injury pain in rats. J Neurosci 32:15843-15848.
Links:














