A New Neuroprotective Docosahexaenoic Acid Preparation Shows Promise for Improved Locomotor Function after Spinal Cord Injury
Posted June 11, 2013
Dr. Adina Michael-Titus, Queen Mary, University of London, United Kingdom
Spinal cord injury (SCI) is a devastating, life-altering condition with major financial and societal impacts. Presently there is no treatment approved by the U.S. Food and Drug Administration that can be administered immediately following SCI to mitigate the progression of damage and thereby improve the functional outcome in the patient. Dr. Adina Michael-Titus has previously shown that omega-3 polyunsaturated fatty acids (PUFAs) are potently neuroprotective when administered acutely following experimental SCI. PUFAs, such as docosahexaenoic acid (DHA), display antioxidant and anti-inflammatory effects and can reduce neuronal and glial cell death following injury.
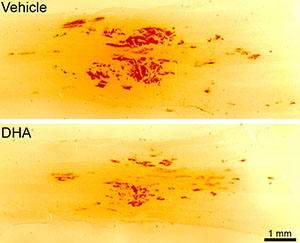
To build on this work, Dr. Michael-Titus received a Fiscal Year 2009 Investigator-Initiated Research Award from the Spinal Cord Injury Research Program to determine the optimum formulation, dosage, and treatment regimen of DHA to improve functional outcomes following experimental SCI. In a mouse contusion model of SCI, Dr. Michael-Titus demonstrated that mice treated with DHA free fatty acid 30 minutes following injury showed significant locomotor improvement beginning 11 days following injury, an effect that was still significant at 5 weeks following SCI. These results were confirmed using a rat contusion model of SCI in which improved locomotor function following DHA treatment was correlated with neuronal survival in the injured spinal cord. There was also evidence of vascular protection following the fatty acid treatment (see Figure, courtesy of Dr Ping Yip). Further, improved locomotion was associated with an increase in microglia and macrophages in the rat model, suggesting an increase in inflammation in the DHA-treated injured spinal cord. This finding is particularly intriguing because it contradicts the hypothesis that improved functional outcome following SCI is associated with reduced inflammation.
Dr. Michael-Titus and her collaborators continue to characterize the protective effect of DHA treatment on experimental SCI and will begin pharmacological experiments to study the tissue distribution and pharmacokinetics of DHA administration, in preparation for human clinical trials. If successful, DHA could provide an efficacious and safe treatment for SCI that could be administered by an emergency response team as soon as possible, limiting the progression of injury and improving the capacity for functional recovery of individuals with SCI.
NovaRed staining was used to demonstrate a significant reduction in hemorrhage following SCI in rats treated with DHA (bottom) compared to those treated with control vehicle (top). Figure courtesy of Dr. Ping Yip.
Links: