Reconstructive Transplant Research
Transfer of Donor Lymph Nodes to Promote Graft Tolerance



Posted August 31, 2021
Dr. Adriano Taddeo, Institute of Virology and Immunology, Switzerland

Dr. Adriano Taddeo
Vascularized composite allotransplantation (VCA) is the transplantation of multiple tissues such as skin, muscle, tendon, nerve, and bone as one unit, like a face or hand. It presents several challenges, particularly the need to regulate the immune system to prevent rejection of the transplanted tissue. The lymphatic system is an important part of the body’s immune system that protects from infection and disease. It consists of tube-like channels called lymphatic vessels that transport immune cells and fluid throughout the body to small bean-shaped masses called lymph nodes (LNs). LNs contain immune cells called B- and T-cells that fight harmful bacteria and viruses, and protect the body. Lymphatic vessels and LNs, along with immune cells, play an important role in regulating the immune response. Transplantation disrupts these lymphatic vessels, and re-establishing these channels is important for the survival of the graft. With the assistance of a 2016 Reconstructive Transplant Research Program Concept Award, Dr. Taddeo and his team found that transferring donor LNs along with the transplanted limb improved graft acceptance and survival by promoting growth of the lymphatic channels.
The team employed a rat hind limb transplantation model (Figure 1) in which recipient rats received grafts either with or without intact donor LNs. Immunosuppressive medication commonly used for survival of the graft was not used in this study. To determine the importance of establishing new lymphatic vessels in the transplanted graft, a third group of recipients with intact LNs also received a blocker called vascular endothelial growth factor receptor-3 -blocker (VEGFR-3 blocker) to inhibit generation of lymphatic channels. This blocker prevents vascular endothelial growth factor C (VEGF-C) from binding to its receptor, thereby preventing the growth and survival of new lymphatic vessels; this process is called lymphangiogenesis.
The results of this study indicate that graft rejection started significantly later in rats transplanted with donor LNs compared to the group that did not include donor LNs (Figure 1). Additionally, the presence of donor LNs increased the levels of factors like VEGF-C that enhance lymphatic growth and regeneration. A variety of inflammatory markers were present in significantly lower levels in rats receiving donor LNs at post-operative day 7 compared to the group without donor LNs, suggesting a delayed inflammatory reaction. Further analyses revealed that donor LN transfer is associated with an increased frequency of donor T cells and donor B cells in the recipient rat. LN transfer increases donor immune cell population, thereby minimizing the recipient’s immune responses to the graft. This may contribute to a favorable outcome for graft survival and can be included in new treatment protocols. In contrast to the rats that received donor LNs, the recipient rats that received both donor LNs and VEGFR-3 blocker to inhibit lymphatic growth showed accelerated graft rejection, suggesting that the beneficial effects of donor LNs depend on active growth of new lymphatic vessels.
Since the investigators did not use immunosuppressive drugs to contain graft rejection, the potential impact of these drugs on the lymphatic system is not known. Additional studies are warranted to understand the role of lymphatics in in conjunction with immunosuppressive therapy. Adding factors known to promote lymphatic survival and regeneration need to be tested in combination with immunosuppressive medications to promote the survival of the graft.
In conclusion, Dr. Taddeo’s team demonstrated that LNs play a pivotal role in the Immune regulation of graft rejection and highlighted the potential of using lymphatic-targeted therapies to promote graft tolerance.
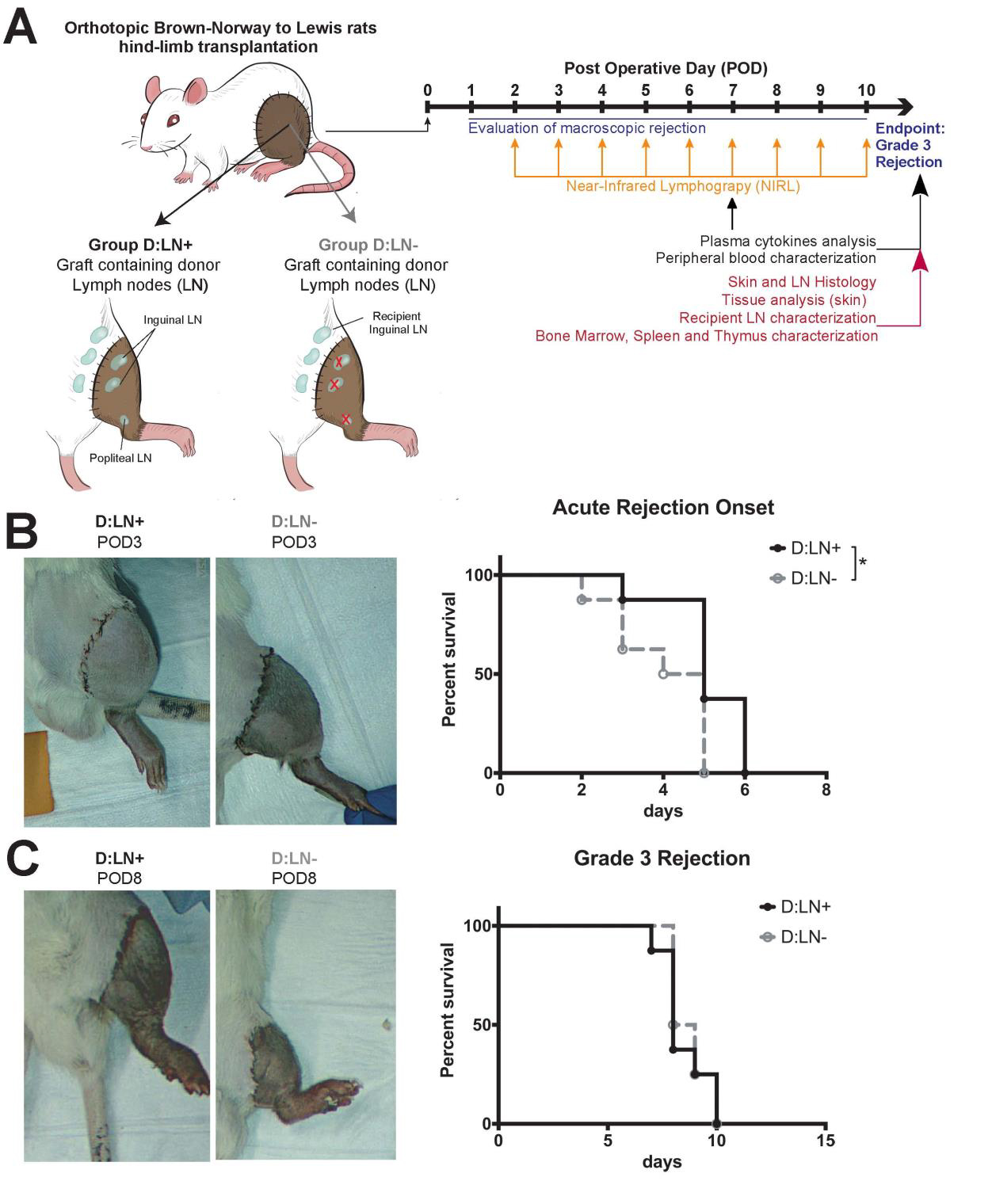 Figure 1: Donor lymph node transfer delay rejection of vascularized composite
Figure 1: Donor lymph node transfer delay rejection of vascularized composite
allotransplants. (A) Schematic representation of the experimental design with recipients receiving either graft containing donor lymph nodes (D: LN+, n=8) or grafts depleted of donor LN (D: LN, n=8) and timeline of the analyses performed. Rejection was graded macroscopically as follows: 0=no rejection, 1=erythema and edema, 2=epidermolysis and exudation, and 3=desquamation, necrosis, and mummification. The rats were sacrificed once grade 3 was reached. (B). Representative pictures of graft rejection at post-operative day (POD) 3 in D: LN+ and D: LN- recipients and onset of acute rejection (i.e., time before showing signs of rejection, reaching Grade 1) represented with Kaplan-Meier survival curves or rejection grading. (C) Representative pictures of graft rejection at POD8 in D: LN+ and D: LN- and overall graft-survival (i.e., time to grade 3 rejection) represented with Kaplan-Meier survival curves. *p<0.05 calculated by Mantel-Cox test.
Reference:
1) Olariu R, Tsai C, Abd El Hafez M, Milusev A, Banz Y, Lese I, Leckenby JI, Constantinescu M, Rieben R, Vögelin E, Taddeo A. Presence of donor lymph nodes within vascularized composite allotransplantation ameliorates vegf-c-mediated lymphangiogenesis and delays the onset of acute rejection. Transplantation. 105 (8):1747-1759. doi: 10.1097/TP.0000000000003601. PMID: 34291766.
Link:
RT160136
Last updated Wednesday, September 17, 2025
 An official website of the United States government
An official website of the United States government
 ) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
