Reconstructive Transplant Research
Project Update
Biomarkers to Predict Rejection in Vascularized Composite Allotransplantation: MMP3



Posted September 24, 2019
Leonardo Riella, M.D., Ph.D.; Bohdan Pomahac, M.D.; Brigham and Women's Hospital, Inc.

Vascularized composite allotransplantation (VCA), the transplantation of multiple tissue types such as a face or hand, has become a viable option to restore function and appearance in some amputees or severely disfigured individuals. However, recipients must adhere to lifelong immunosuppression regimens, and more than 80% of recipients will experience an episode of acute rejection within the first year (1). Novel non-invasive biomarkers for the diagnosis of rejection in VCA are needed as an alternative to the current reliance on skin biopsies and the Banff classification system. Skin biopsy can cause additional morbidity for the patient, including scarring, bleeding, or infection, and the Banff grading system is a semi-quantitative method that can be subject to variability. Therefore, identification of biomarkers that could be monitored non-invasively via blood samples is needed to better manage VCA patient care and predict and monitor rejection episodes.
In March 2019, the Reconstructive Transplant Research Program highlighted the project of Drs. Leonardo Riella and Bohdan Pomahac at Brigham and Women’s Hospital, which sought to identify novel non-invasive biomarkers of allograft rejection in VCA recipients (https://cdmrp.health.mil/rtrp/research_highlights/18riella_highlight). We reported then that the team had examined the gene expression profile from skin biopsy samples of a single face transplant recipient and identified 79 genes upregulated during rejection episodes compared to periods of non-rejection.
Since then, the team has gone on to analyze serum samples from six face transplant recipients collected over a follow-up range of 12-54 months, representing episodes of no-rejection, non-severe rejection, and severe rejection (2). Using a novel proteomic platform called SOMAscan that uses modified aptamers to bind to proteins and quantify their expression levels, a signature of five proteins capable of discriminating severe rejection from no-rejection and non-severe rejection was identified. MMP3 (Matrix Metalloproteinase 3), ACY1 (Aminoacylase-1), IL1R2 (Interleukin-1 receptor type 2), SERPINA4 (Kallistatin), and CPB2 (Carboxypeptidase B2) were found to be significantly upregulated during severe rejection. MMP3 showed the highest levels of upregulation and was further validated using ELISA, a standard immunoassay technology. The high correlation between MMP3 levels seen between the two different platforms demonstrates, for the first time in the field of transplantation, that SOMAscan accurately reflects the relative protein levels in a sample (Figure 1A). Using the ELISA data, a plot demonstrating the diagnostic ability of MMP3 to discriminate between no-rejection and severe rejection, as well as non-severe from severe rejection, was generated (Figure 1B).
These findings are the first to identify MMP3 as a potential noninvasive biomarker for monitoring severe rejection episodes, which require strong systemic immunosuppression. Additional validation is needed with a larger patient population including other VCA types and across additional clinical centers. With additional studies, this may lead to better understanding of the molecular process underlying rejection as well as better methods of detecting and preventing rejection.
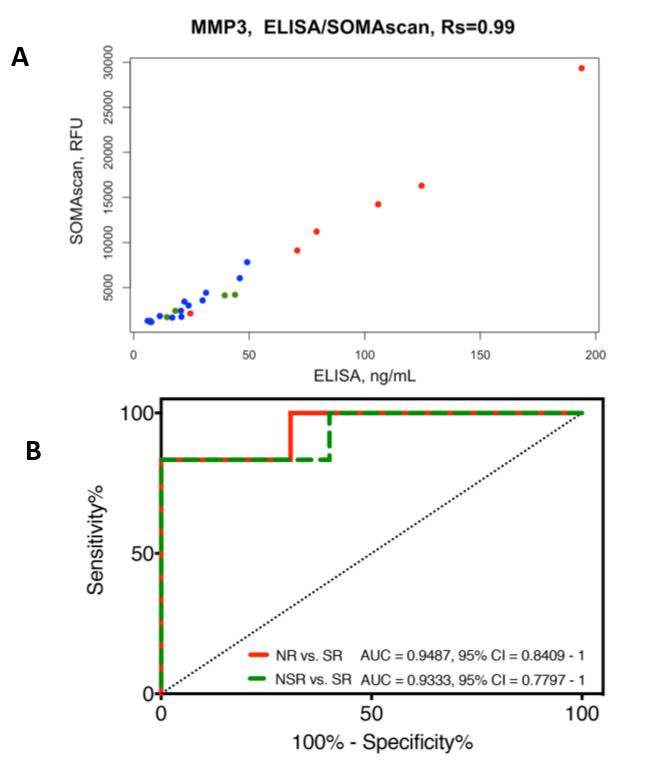
Figure 1A. Correlation between SOMAscan and ELISA measurements of MMP3 serum levels. High correlation (Rs = 0.99) was found between SOMAscan and ELISA data for the MMP3 protein. Severe rejection (n = 6) is shown in red; non-severe rejection (n = 5) in green; and no-rejection (n = 13) in blue. RFU = relative fluorescence units; Rs = Spearman’s rank correlation coefficient.
Figure 1B. Receiver operating characteristic (ROC) curves of the MMP3 ELISA data between the no-rejection, non-severe rejection, and severe rejection samples. MMP3 shows good performance as a diagnostic marker for severe rejection (SR) episodes compared to no-rejection (NR; AUC = 0.9487; 95% CI 0.8409 to 1) and non-severe rejection (NSR; AUC = 0.9333; 95% CI 0.7797 to 1) episodes.
References:
1. Kueckelhaus M, Fischer S, Seyda M, Bueno EM, Aycart MA, Alhefzi M, ElKhal A, Pomahac B, Tullius SG. Vascularized composite allotransplantation: current standards and novel approaches to prevent acute rejection and chronic allograft deterioration. Transpl Int. 2016 Jun;29(6):655-62. doi: 10.1111/tri.12652. Epub 2015 Sep 14. Review. PubMed PMID: 26265179; PubMed Central PMCID: PMC4785085.
2. Kollar B, Shubin A, Borges TJ, Tasigiorgos S, Win TS, Lian CG, Dillon ST, Gu X, Wyrobnik I, Murphy GF, Pomahac B, Libermann TA, Riella LV. Increased levels of circulating MMP3 correlate with severe rejection in face transplantation. Sci Rep. 2018 Oct 8;8(1):14915. doi: 10.1038/s41598-018-33272-7. PubMed PMID: 30297859; PubMed Central PMCID: PMC6175842.
Last updated Friday, December 13, 2024














