Parkinson's
Parkinson's Risk Estimation Using Digital Diagnosis Codes and Treatments



Posted June 6, 2023
Brad A. Racette, M.D., Washington University School of Medicine in St. Louis and Barrow Neurological Institute, Phoenix
Susan Searles Nielsen, Ph.D., Washington University School of Medicine in St. Louis
 Dr. Brad A. Racette
Dr. Brad A. Racette
(Photo Provided)
 Dr. Susan Searles Nielsen
Dr. Susan Searles Nielsen
(Photo Provided)
In North America, more than one million people are affected by a progressive neurodegenerative disease called Parkinson's disease (PD).1 PD is largely diagnosed when patients develop classic motor features, such as resting tremor, rigidity (muscle stiffness), slow movement, and postural instability. However, many subtle motor and non-motor symptoms predate these, a period called the 'prodromal period.' These subtle motor findings result in falls, fractures, and traumatic brain injury. Non-motor symptoms include cognitive dysfunction, sleep disruptions, and fatigue. The prodromal period may represent a critical time point to the prognosis of PD because providers could identify the PD patients earlier. Early diagnosis of PD could provide the opportunity to avoid significant neurological damage 2 and prevent fall-related outcomes, such as traumatic brain injuries3 and fractures.4 Beginning more than five years before PD diagnosis, fall-related traumas occur more frequently in undiagnosed PD patients than comparable individuals without PD.4
Drs. Racette and Nielsen and their team previously developed a predictive model for detecting PD during the prodromal period, using only demographic and Medicare claims data, including diagnosis and procedure codes data from the International Classification of Diseases version 9 (ICD9) for up to five years prior to PD diagnosis5 (see Figure 1). Since PD is an age-related disease, Medicare data is an ideal source of population-based data to predict which patients will be diagnosed with PD. With a fiscal year 2019 (FY19) Investigator-Initiated Research Award, Drs. Racette and Nielsen were funded by the Neurotoxin Exposure Treatment Parkinson's (NETP) Program to continue their work on a predictive model to identify which patients are at the highest risk of developing PD in the next three years. In FY22, the NETP was transitioned by Congress to the Parkinson's Research Program (PRP), which now manages this award.
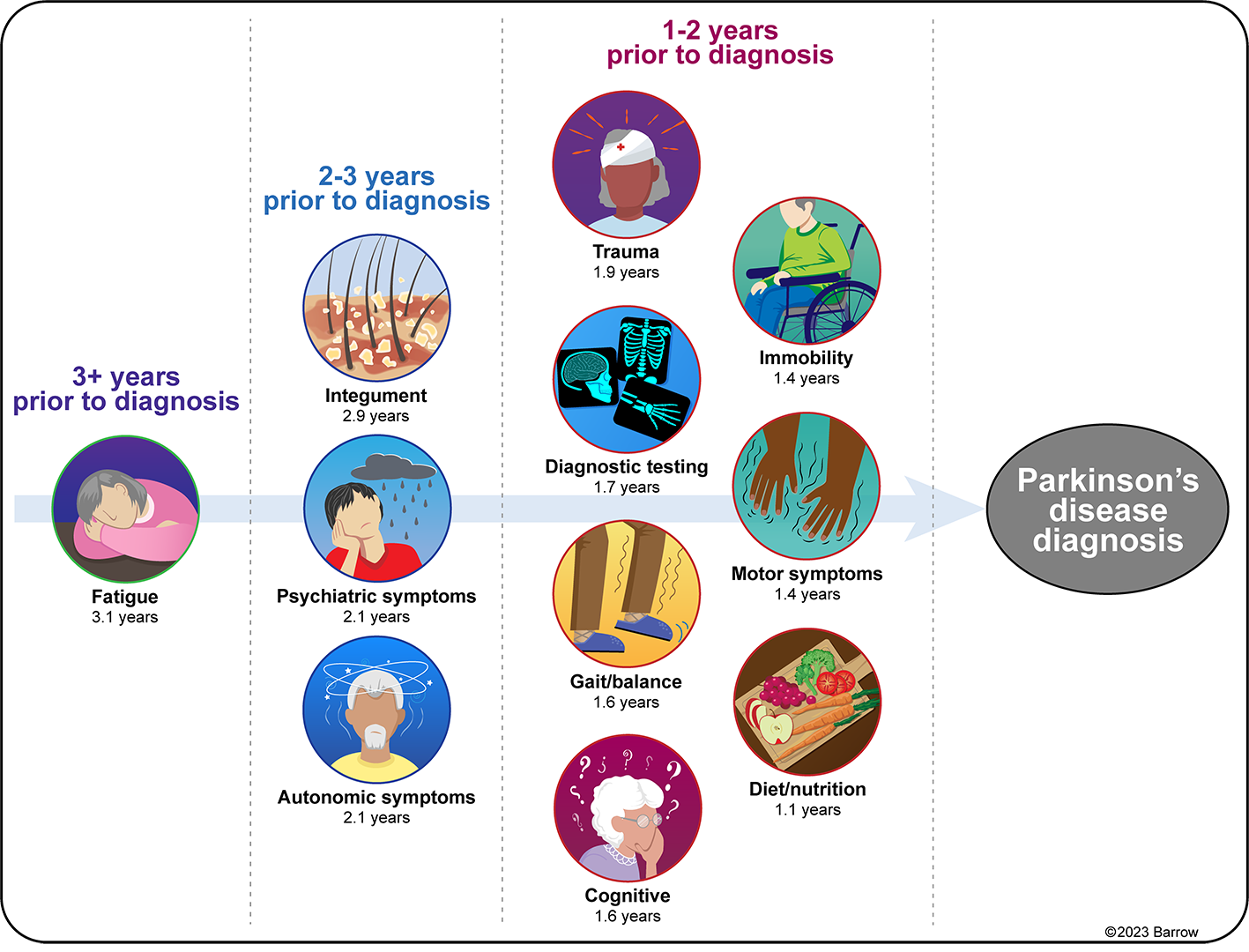 Figure 1: Selected types of diagnosis and procedure codes can help predict a diagnosis of Parkinson's disease (PD) within five years. The number of years before PD diagnosis that these predictive codes first appear in Medicare claims varies according to the type of code. Codes for fatigue first occur more than 3.1 years prior to PD diagnosis for half of PD patients, autonomic symptoms more than 2.1 years prior, traumas more than 1.9 years prior, and motor symptoms more than 1.4 to 1.6 years prior, for example. A variety of diagnostic tests also begin occurring a median of 1.7 years prior to PD diagnosis. (Figure Provided)
Figure 1: Selected types of diagnosis and procedure codes can help predict a diagnosis of Parkinson's disease (PD) within five years. The number of years before PD diagnosis that these predictive codes first appear in Medicare claims varies according to the type of code. Codes for fatigue first occur more than 3.1 years prior to PD diagnosis for half of PD patients, autonomic symptoms more than 2.1 years prior, traumas more than 1.9 years prior, and motor symptoms more than 1.4 to 1.6 years prior, for example. A variety of diagnostic tests also begin occurring a median of 1.7 years prior to PD diagnosis. (Figure Provided)
This study, which has already led to two publications6,7 in the scientific journal PLoS ONE, includes work that further enhances the team's previous work by applying sophisticated machine learning methods to predict PD. By acquiring ICD version 10 (ICD10) data from the Centers for Medicare and Medicaid Services (CMS) for a newer and larger study, the computer model to predict PD needed to be redeveloped, because there were four times as many codes added between ICD9 and ICD10 versions. The research team also considered whether the addition of Medicare Part D prescription medication data improved their ability to identify patients. The Part D data also allowed the team to explore whether any medications were associated with a lower risk of developing PD, to identify potentially neuroprotective medications that are already U.S. Food and Drug Administration (FDA) approved for other medical conditions. The team hypothesized that this predictive model will help identify people who will develop PD, using only basic demographic and Medicare medical claims data 3-5 years prior to disease diagnosis. They also hypothesized the Part D prescription medication data would enable them to identify medications that might be neuroprotective. Such findings would make the PD predictive model even more useful because any identified neuroprotective medications might be more effective when started earlier in the course of PD.
In their 2021 study, Drs. Racette and Nielsen and their team of investigators identified U.S. residents aged 66-90, who relied solely on Medicare in 2009. Since Medicare is the only nationwide health insurance coverage universally available in the U.S., the study was restricted to the eligible population of those 65 and older. They included all incident (newly diagnosed) PD cases and a random sample of comparable beneficiaries as controls. A PD case was determined from Part A and B Medicare claims data for 2004-2009 by having at least one ICD9 code for PD in 2009, but in no prior year of claims. The controls for the study met the same study eligibility criteria; however, they had no ICD9 code for PD. The team linked their detailed Medicare claims files for five years prior to the PD diagnosis or control reference date. They constructed three models to analyze these data: 1) penalized regression without prescription medications, 2) penalized regression with prescription medications, and 3) a random forest classifier (machine learning) that considered the prescription medications as part of the model. They also experimented with combining both approaches to produce a single combined classifier by fitting a penalized logistic regression model that also used the probability of PD generated by the final random forest as a predictor. They then used standard statistical measures to assess model performance.
Model performance was similar across the three models. However, adding the random forest classifier to the penalized regression model slightly improved the model performance because the random forest model was able to identify many unique codes associated with PD as compared to the penalized regression models.6 Nonetheless, there was high correlation between respective model predictions for detecting PD, which further validates the previous model that the team generated.5
Drs. Racette and Nielsen also used the same PD data and similar data for patients with Alzheimer's disease or amyotrophic lateral sclerosis (ALS) to identify a type of medication that might be neuroprotective across all three diseases. The team found that medications that block a particular biological pathway in humans were associated consistently with a reduced risk of all three neurodegenerative diseases.7 If these medications and pathway are confirmed to slow progression of neurodegenerative diseases, then this additional work will further enhance the usefulness of identification of PD earlier in the course of the prodromal period via the team's PD predictive model.
Researchers have been working to determine how to diagnose patients with PD during the prodromal period, as well as to study the effect of medications that could potentially slow disease progression. This ICD10-based predictive model to be developed in this study is the logical next step. Ultimately, developing a final computer model that is ready to be implemented using contemporary Medicare claims data will give clinicians the ability to identify patients with undiagnosed PD and intervene earlier to treat symptoms to reduce morbidity and mortality due to falls, fractures, and traumatic brain injury.
References:
1"Who Has Parkinson's?" Parkinson's Foundation, https://www.parkinson.org/understanding-parkinsons/statistics.
2Rees RN, Acharya AP, Schrag A, and Noyce AJ. 2018. An early diagnosis is not the same as a timely diagnosis of Parkinson's disease. F1000 Research 7:F1000 Faculty
3Camacho-Soto A, Warden MN, Searles Nielsen S, et al. Traumatic brain injury in the prodromal period of Parkinson's disease: A large epidemiological study using Medicare data. Annals of Neurology 2017;82:744-754. PMID: 29024046 PMCID: PMC5812286 doi: 10.1002/ana.25074.
4Camacho-Soto A, Gross A, Searles Nielsen S, et al. Fractures in the prodromal period of Parkinson disease. Neurology 2020; 94:e2448-e2456.
5Searles Nielsen S, Warden MN, Camacho-Soto A, et al. A predictive model to identify Parkinson disease from administrative claims data. Neurology 2017;89:1448-1456.
6Warden MN, Searles Nielsen S, Camacho-Soto A, Garnett R, and Racette BA. 2021. A comparison of prediction approaches for identifying prodromal Parkinson disease.
7Song Y, Racette BA, Camacho-Soto, A, and Searles Nielsen S. Biologic targets of prescription medications and risk of neurodegenerative disease in United States Medicare beneficiaries. PLoS ONE, 2023; 18(5):e0285011. PMID: 37195983 PMCID: PMC10191296 doi: 10.1371/journal.pone.0285011.
Last updated Wednesday, September 17, 2025
 An official website of the United States government
An official website of the United States government
 ) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
