Parkinson's
Investigator-Initiated Research Award Highlights



Posted June 7, 2023
Jason Cannon, Ph.D., Purdue University (mPI)
Ellen Wells, Ph.D., Purdue University (mPI)
 Dr. Jason Cannon
Dr. Jason Cannon
(Photo Provided)
 Dr. Ellen Wells
Dr. Ellen Wells(Photo Provided)
While research into the causes of Parkinson's disease (PD) has focused extensively on neurological trauma and exposure to toxins, such as chemical warfare agents and heavy metals, the neurodegenerative effects of substances commonly encountered during military service have been less well studied, such as the organophosphate (OP) pesticide chlorpyrifos. With the help of an Investigator-Initiated Research Award, Drs. Jason Cannon (toxicology) and Ellen Wells (epidemiology) are co-leading a highly interdisciplinary team of researchers (including researchers at Purdue University, the Indianapolis Veterans Affairs, and the University of Iowa) seeking to better understand how chlorpyrifos may increase the risk of PD. Their team will use human, rat, and cellular studies to test the hypothesis that OP exposure among military Veterans is associated with PD and determine whether chlorpyriphos exposure in a military setting damages dopamine-generating neurons in the brain, which can cause neurological and psychological symptoms that require medical treatment. This study will not only shed light on the correlation between chlorpyrifos exposure and development of PD but will also inform clinical monitoring guidelines for military personnel who have been exposed to chlorpyrifos, as well as strategies to limit future military exposures.
Richard Gordon, Ph.D., Queensland University of Technology, Australia
 Dr. Richard Gordon
Dr. Richard Gordon(Photo Provided)
Uncontrolled inflammation has been shown to cause and accelerate damage dopamine-producing neurons and drive alpha synuclein aggregation, which are central hallmarks of Parkinson's disease (PD). Researchers believe that modulating the body's immune and inflammatory responses could interrupt these harmful processes and potentially slow disease progression. Neuroinflammation expert Associate Professor Richard Gordon will apply his Investigator-Initiated Research Award to expand on his previous research, assessing a novel Nod-like Receptor (NLR) family protein that is involved in regulation of inflammation. Research led by Dr. Gordon's team found this pathway is altered in people with PD as well as in disease models. This research program will use samples from Parkinson's patients, animal models, and cell culture studies to understand the role of this novel NLR pathway in PD.
https://research.qut.edu.au/cmr/
twitter.com/Gordon_NeuroLab
Yoon-Seong Kim, M.D., Ph.D., RWJMS-Institute for Neurological Therapeutics, Rutgers
 Dr. Yoon-Seong Kim
Dr. Yoon-Seong Kim(Photo Provided)
There is growing evidence to support the hypothesis that Parkinson's disease (PD) may begin in the gut microbiome, where aggregations of the protein
https://yoonkimlab.com/
Links:
A Cell Type-Specific Epigenetic and Transcriptional Profile of the Development of L-DOPA-Induced Dyskinesia in a Neurotoxin Model of Parkinson Disease
Public and Technical Abstracts: The Role of NADPH Oxidase1 (NOX1) in the Colon in Rotenone-Induced a-Synuclein Pathology Propagation Along the Gut-Brain Axis
Ken Nakamura, M.D., Ph.D., J. David Gladstone Institutes
 Dr. Ken Nakamura
Dr. Ken Nakamura
(Photo Provided)
Accumulating evidence suggests that insufficient production of the energy carrying compound adenosine triphosphate (ATP) in the mitochondria of cells plays a role the development of Parkinson's disease (PD). However, researchers still do not understand how energy deficiency contributes to PD. With the support of an Investigator-Initiated Research Award, Dr. Ken Nakamura, an expert in mitochondrial biology, will use cell cultures of human neurons to test his innovative hypothesis that energy failure due to insufficient mitochondrial ATP production promotes the aggregation and spread of the central PD protein a-synuclein in neurons. This research could lead to new metabolism-based therapies that inhibit a-synuclein aggregation and spread, thereby slowing the progression of PD.
https://labs.gladstone.org/nakamura/
Christine Nguyen, Ph.D., The University of Melbourne
 Dr. Christine Nguyen
Dr. Christine Nguyen(Photo Provided)
The ability to measure the overaccumulation of a-synuclein protein in patients - a hallmark of Parkinson's disease (PD) - would be an invaluable diagnostic tool for clinicians seeking to assess disease progression and treatment efficacy in real time. Currently, there is no low-risk, non-invasive way to accomplish this. In a paradigm-shifting approach to the problem, ocular biomarker expert Dr. Christine Nguyen proposes to use her Investigator-Initiated Research Award to adapt existing multimodal retinal imaging technologies to detect a-synuclein accumulations in the eye. Her research involves testing three types of retinal imaging markers on mouse models that mimic environmental and genetic risk factors to assess their ability to detect retinal
https://findanexpert.unimelb.edu.au/profile/25627-christine-nguyen
Link:
Public and Technical Abstracts: Imagine Imaging Toxin-Mediated Alpha-Synuclein in the Eye
Clemens Scherzer, M.D., Harvard Medical School, Brigham and Women's Hospital
 Dr. Clemens Scherzer
Dr. Clemens Scherzer(Photo Provided)
 Figure 1: Can genome-wide polygenic scores and health data be used to predict (and prevent) disease progression in Parkinson's patients? (Figure Provided)
Figure 1: Can genome-wide polygenic scores and health data be used to predict (and prevent) disease progression in Parkinson's patients? (Figure Provided)
Identifying variations of individual genes is insufficient in determining whether a person is at risk of developing Parkinson's disease (PD). However, if researchers can compile a complete set of variations associated with PD across the entire human genome, the resulting polygenic score – the total number of genetic variants that, taken together, can be used to estimate disease risk – could prove to be a useful predictor of an individual's risk of developing dementia, one of PD's most debilitating manifestations. With support from an Investigator-Initiated Research Award,
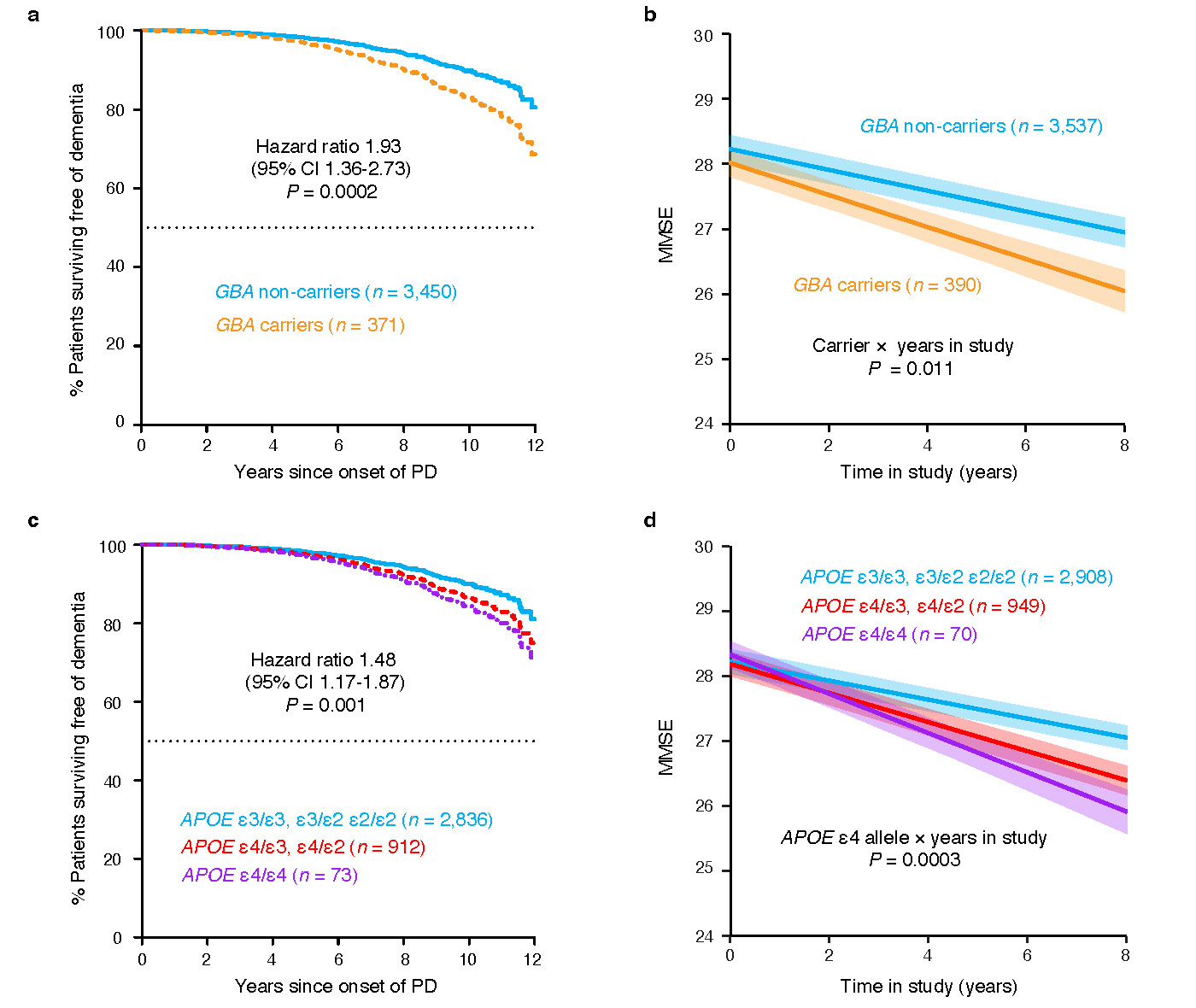 Figure 2: GBA and APOE gene variants are linked to fast disease progression (a,b). Memory scores decline more rapidly in patients carrying variants in these genes. Images from Liu et al., Nature Genetics, 2021. (Figure Provided)
Figure 2: GBA and APOE gene variants are linked to fast disease progression (a,b). Memory scores decline more rapidly in patients carrying variants in these genes. Images from Liu et al., Nature Genetics, 2021. (Figure Provided)
Link:
Public and Technical Abstracts: Polygenic Score for Dementia in Parkinson's Disease
Ronald Tjalkens, Ph.D., Colorado State University
 Dr. Ronald Tjalkens
Dr. Ronald Tjalkens
(Photo Provided)
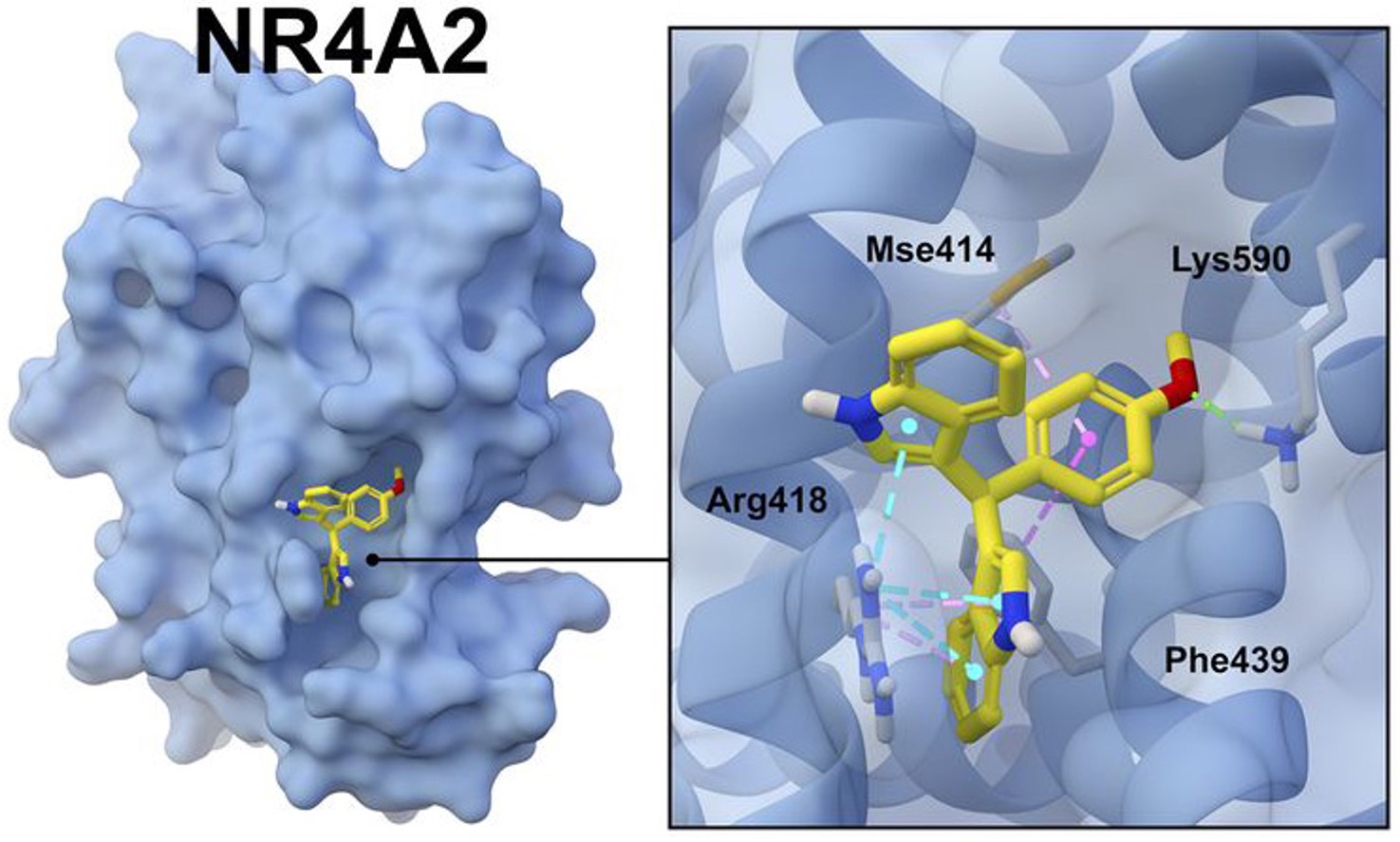 Figure 3: The nuclear receptor NR4A2/Nurr1 is key protein regulating inflammation in the supporting cells of the brain, called glial cells. When exposed to neurotoxic environmental chemicals, glial cells turn on inflammatory genes that can damage neurons over time. NR4A2 is an important "off switch" for these genes. Research in the Tjalkens laboratory is studying how novel small molecules can be developed as therapeutic agents to activate the anti-inflammatory activity of NR4A2 to protect neurons from neurotoxic injury. The long-term goal of this CDMRP/NETP project is identify the best compounds to slow down the progression of Parkinson's-like symptoms in individuals exposed to neurotoxic agents. (Figure Provided)
Figure 3: The nuclear receptor NR4A2/Nurr1 is key protein regulating inflammation in the supporting cells of the brain, called glial cells. When exposed to neurotoxic environmental chemicals, glial cells turn on inflammatory genes that can damage neurons over time. NR4A2 is an important "off switch" for these genes. Research in the Tjalkens laboratory is studying how novel small molecules can be developed as therapeutic agents to activate the anti-inflammatory activity of NR4A2 to protect neurons from neurotoxic injury. The long-term goal of this CDMRP/NETP project is identify the best compounds to slow down the progression of Parkinson's-like symptoms in individuals exposed to neurotoxic agents. (Figure Provided)
Exposure to environmental toxins like pesticides increases the risk of developing Parkinson's disease (PD) due to exposure-associated chronic inflammation. The ability to inhibit inflammation, therefore, could potentially help slow disease progression. Dr. Ronald Tjalkens is applying his Investigator-Initiated Research Award to explore how synthetic versions of molecules that bind to cellular receptors (called ligands) might be used to modulate the inflammation-inhibiting characteristics of NR4A/Nurr1, a specific receptor protein in neuron support cells (called glial cells), after exposure to the pesticide rotenone. His team will begin by testing a range of synthetic ligands for their ability to bond strongly with NR4A/Nurr1 and to modulate rotenone-triggered inflammation in glial cells and neurons, then characterize the properties of the most effective of those ligands for further development as potential therapies for treating PD associated with environmental exposure.
Last updated Friday, December 19, 2025
 An official website of the United States government
An official website of the United States government
 ) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
