Parkinson's
The PRP Presents the Fiscal Year 2022 Early Investigator Research Award Recipients



Posted December 19, 2023
Neurocircuitry of Impaired Visual and Memory Integration in Parkinson's Disease
 Dr. Leila Montaser Kouhsari
Dr. Leila Montaser Kouhsari
(Photo Provided)
Leila Montaser Kouhsari, M.D., Ph.D., Brigham and Women Hospital, Harvard University
Cognitive impairments in PD can affect the parts of the brain involved in processing visuospatial tasks and memory, leading to an increased risk of dementia, falls, and hallucinations. With support from an Early Investigator Research Award, Leila Montaser Kouhsari, M. D., Ph.D., hopes to identify structural and functional changes in the brain when people with PD perform memory-based visuospatial decision-making tasks. A better understanding of how the brain integrates visual information with learned information from memories could clarify the decision-making impairment among people with PD. This, in turn, may lead to the discovery of biomarkers that clinicians can use to predict falls, dementia, and hallucinations.
Microglia and NLRP3 Inflammasome Contribution to Nonmotor Deficits in Parkinson's Disease
Nikhil Panicker, Ph.D., Cleveland Clinic Foundation
 Dr. Nikhil Panicker
Dr. Nikhil Panicker
(Photo Provided)
Dementia can significantly impact the quality of life of PD patients and their caregivers. In fact, dementia is the primary reason people with PD are admitted to long-term care.1 Elucidating the mechanisms driving PD-dementia could enable clinicians to slow disease progression, improve quality of life, and reduce the burden of care. With support from an Early Investigator Research Award, Nikhil Panicker, Ph.D., is investigating the relationship between two classic signs of PD: the aggregation of a protein known as alpha-synuclein, or a-Syn, and the inflammation of immune cells, within the brain. Recent studies have demonstrated that a-Syn can spread and accumulate throughout the brain. Misfolded a-Syn can act as a template, corrupting endogenous a-Syn and spreading pathology across the brain. Preliminary work performed by Dr. Panicker shows that a-Syn controls the hyperactivation of a particular gene associated with the inflammation process, NLRP3, within immune cells in the brain called microglia. Identifying the process through which hyperactivation of NLRP3 contributes to a-Syn propagation may suggest new therapeutic targets for addressing and minimizing the effects of PD-related dementia.
Reference:
1Parkinson's Disease: Challenges, Progress, and Promise. Reviewed 2023. National Institutes of Health.
https://www.ninds.nih.gov/current-research/focus-disorders/parkinsons-disease-research/parkinsons-disease-challenges-progress-and-promise
From Dreams to Degeneration: Sleep-Wake Disturbances as a Target for Novel Biomarkers and Therapies for Patients with Parkinson's Disease
Elie Matar, Ph.D., University of Sydney
 Dr. Elie Matar
Dr. Elie Matar
(Photo Provided)
Sleep disturbances are a hallmark of PD. Originally, researchers believed them to be a byproduct of worsening motor symptoms or side effects of medications. However, sleep problems are now recognized as part of the disorder and significantly affect quality of life of patients.1 They can often occur early and even precede motor symptoms, as with rapid-eye-movement sleep behavior disorder which is now one of the most powerful clinical predictors for developing PD.2 Others have shown in animals that poor sleep can promote the buildup of toxic proteins in the brain, raising the possibility that sleep problems can also contribute to or worsen the disease.3 Elie Matar's study, supported by an Early Investigator Research Award, will try to unravel the relationship between sleep and PD, by looking at post-mortem samples together with brain imaging and electrophysiological recordings in patients to understand the mechanisms of sleep disturbances how they relate to PD progression. Because sleep disturbances have the potential to impact a variety of motor and non-motor symptoms of PD, a better understanding the mechanisms of sleep disturbances could lead to identifying new therapeutic targets for treating sleep disorders and even limit disease progression.
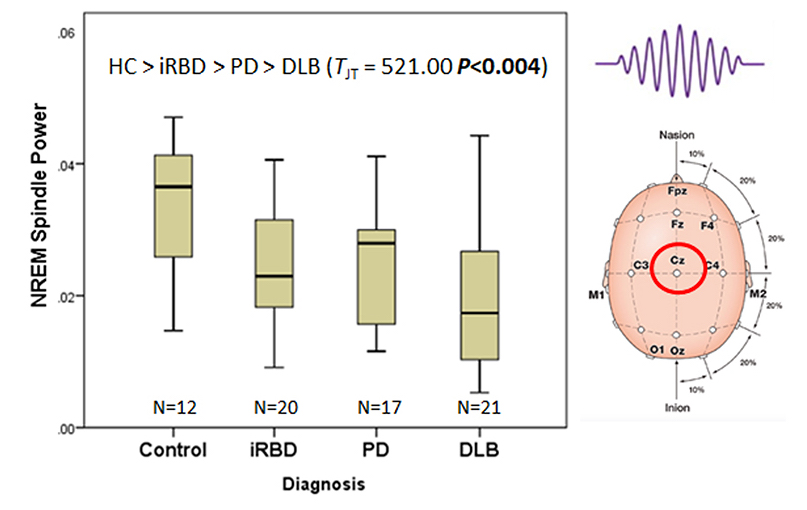 Figure 1: Early data showing progressive changes in one of the electrical signatures of sleep called "sleep spindles" with advancing Parkinson's disease pathology.
Figure 1: Early data showing progressive changes in one of the electrical signatures of sleep called "sleep spindles" with advancing Parkinson's disease pathology.
References:
1Ylikoski A, Martikainen K, Sieminski M, and Partinen M. 2017. Sleeping difficulties and health-related quality of life in Parkinson's disease. Acta Neurol Scand. 135(4):459-468. https://doi.org/10.1111/ane.12620
2Postuma RB, et al. Risk and predictors of dementia and parkinsonism in idiopathic REM sleep behaviour disorder: a multicentre study. Brain. 2019 Mar 1;142(3):744-759.
https://pubmed.ncbi.nlm.nih.gov/30789229/
3Bohnen NI, Hu MTM. Sleep Disturbance as Potential Risk and Progression Factor for Parkinson's Disease. J Parkinsons Dis. 2019;9(3):603-614.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6700634/
Neurophysiology Biomarkers of Cognitive Impairment Associated with Deep Brain Stimulation
 Dr. Sarah Bick
Dr. Sarah Bick
(Photo Provided)
Sarah Bick, M.D., Vanderbilt University Medical Center
Deep brain stimulation, or DBS, a surgical intervention that stimulates the subthalamic nucleus region of the brain, is used for the treatment of several Parkinson's disease, or PD, symptoms. However, DBS can also cause cognitive impairment in some patients. With support from an Early Investigator Research Award, Sarah Bick, M.D., aims to build on her previous research into beta waves - high-frequency brain waves associated with conscious thought - to identify neurological "signatures" associated with DBS-related cognitive impairment. These signatures could be used to identify people who are at risk of experiencing cognitive impairment resulting from DBS treatment and to adapt treatment to minimize this side effect.
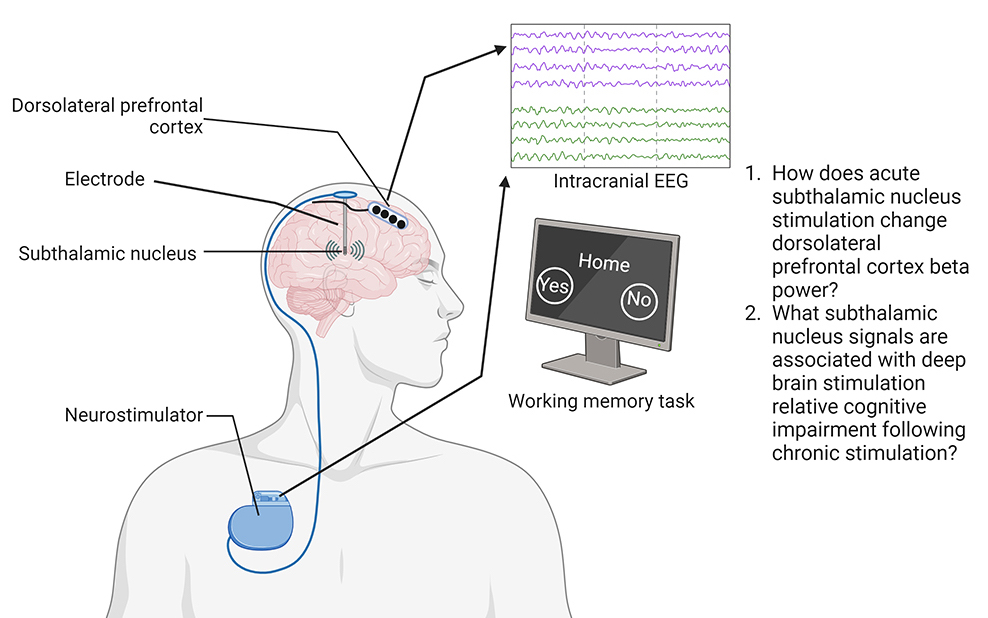 Figure 2: Experimental design. Intracranial neural signals are recorded while subjects participate in a working memory task both with stimulation on and with stimulation off in order to identify neural biomarkers of deep brain stimulation related cognitive impairment. (Diagram Provided)
Figure 2: Experimental design. Intracranial neural signals are recorded while subjects participate in a working memory task both with stimulation on and with stimulation off in order to identify neural biomarkers of deep brain stimulation related cognitive impairment. (Diagram Provided)
Last updated Friday, December 12, 2025
 An official website of the United States government
An official website of the United States government
 ) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
