Parkinson's
Early Investigator Research Award Highlights



Posted May 19, 2023
Avraham Ashkenazi, Ph.D., Tel Aviv University
 Dr. Avraham Ashkenazi
Dr. Avraham Ashkenazi (Photo Provided)
Dr. Avraham Ashkenazi's research focuses on developing new diagnostic and therapeutic techniques for Parkinson's disease (PD) by identifying and understanding the cellular mechanisms that enable neurons to survive. With support from an Early-Investigator Research Award, Dr. Ashkenazi plans to use stem cell technology to explore how exposure to environmental toxins and certain types of proteins affect peripheral autonomic neurons, which are responsible for the functioning of the heart, bladder, intestines, and blood vessels. Because many PD patients will develop gastrointestinal, cardiovascular, or even psychiatric disorders before they experience the onset of motor symptoms, a better understanding of dysfunction in peripheral autonomic neurons not only could help physicians to predict and diagnose PD earlier, but also could lead to the development of novel therapeutic treatment strategies.
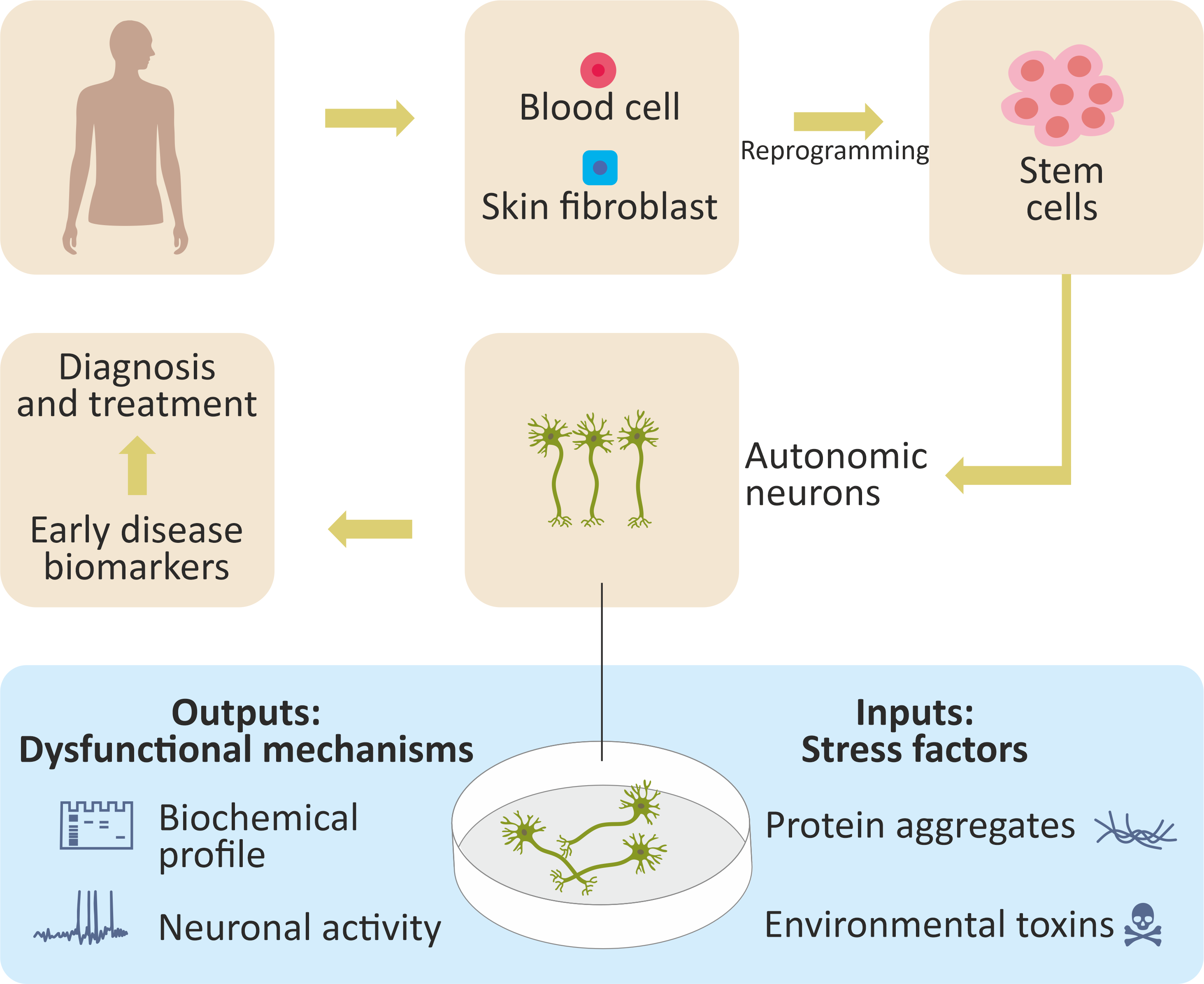 Figure 1: Pluripotent stem cell technology to study the autonomic nervous system in Parkinson's disease. (Figure Provided)
Figure 1: Pluripotent stem cell technology to study the autonomic nervous system in Parkinson's disease. (Figure Provided)
Margaret Caulfield, Ph.D., Michigan State University
 Dr. Margaret Caulfield
Dr. Margaret Caulfield (Photo Provided)
The success of cell-based therapies in treating Parkinson's disease (PD), such as grafting new dopamine (DA) neurons into the brain of a person with PD, can vary widely from patient to patient. Dr. Margaret Caulfield's study, supported by an Early-Investigator Research Award, seeks to better understand how an individual's unique genetics impacts the success of DA cell replacement therapy. Focusing on a specific genetic variation called rs6265 (also called Val66Met), Dr. Caulfield will test how the presence of the rs6265 gene variant, which suppresses the release of brain-derived neurotrophic factor (BDNF), a growth factor that is critical for normal neuron development and function, influences therapeutic outcome of grafting. Curiously, this rs6265 variation has both clinically positive and negative influences on DA cell therapy. Ultimately, Dr. Caulfield seeks to determine whether "the positive" associated with the rs6265 variation is related to a unique factor that improves the likelihood that DA cell replacement therapy will be successful. She also will examine whether supplementing levels of the BDNF in rs6265 subjects can mitigate "the negative." Understanding how to harness the positive aspects, while modifying the negative, will provide for optimized therapeutic development for PD.
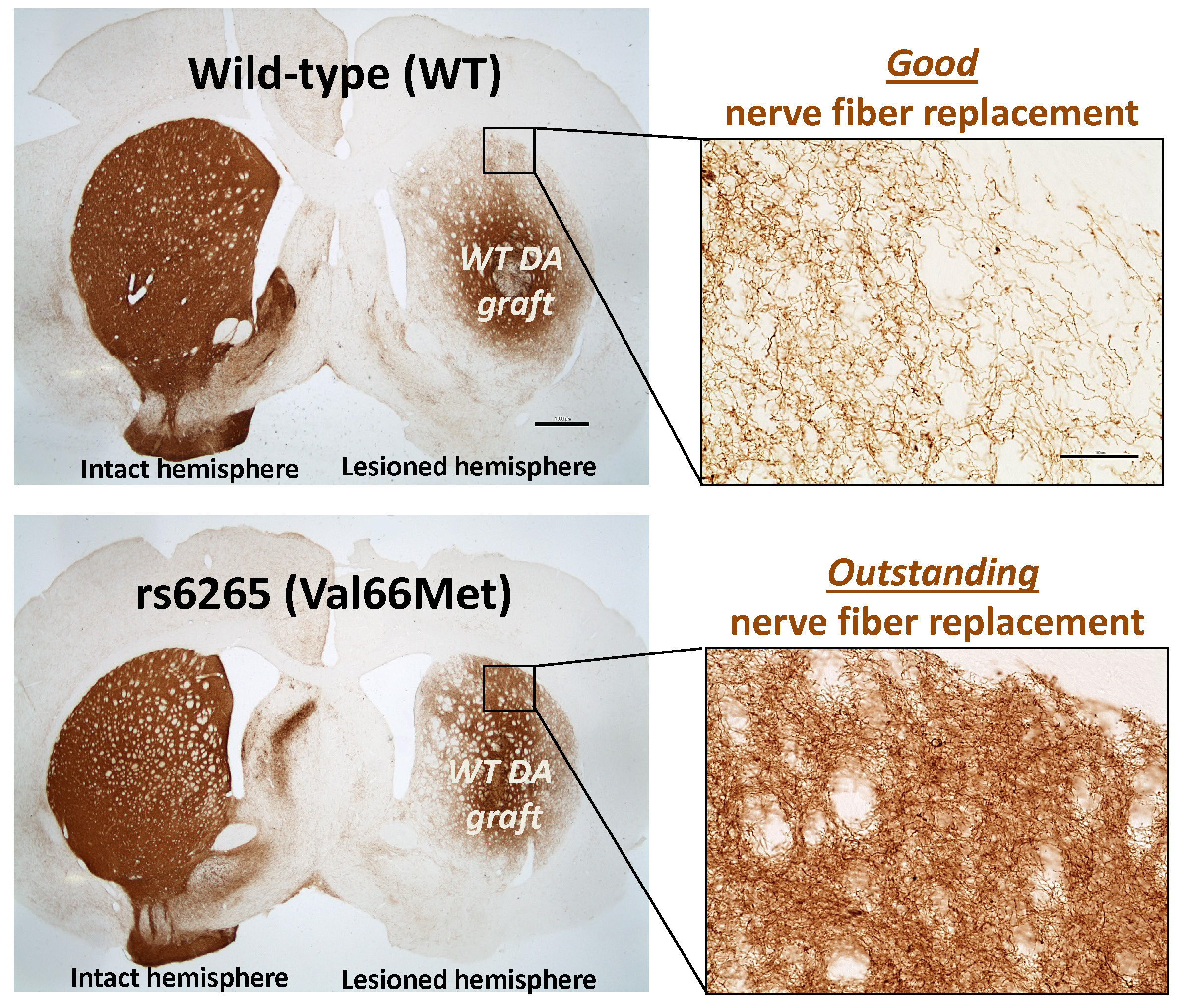 Figure 2: Wild type dopamine cell replacement into
Figure 2: Wild type dopamine cell replacement into
Karen L. Eskow Jaunarajs, Ph.D., University of Alabama at Birmingham
 Dr. Karen L. Eskow Jaunarajs
Dr. Karen L. Eskow Jaunarajs
Levodopa (L-DOPA) treatment is one of the few effective drug therapies for Parkinson's disease (PD), but long-term use of this treatment leads to abnormal uncontrollable movements and postures, called L-DOPA-induced dyskinesia and dystonia (LIDD). Dr. Karen Jaunarajs studies the role that the striatum - the brain's primary motor area - plays in movement disorders. With the help of an Early-Investigator Research Award, she seeks to understand how L-DOPA treatment affects gene expression and transcription in individual cells in the striatum by affecting the three-dimensional availability of DNA as LIDD develops. A more complete understanding of how gene expression changes are affected by L-DOPA and LIDD could lead to the development of new therapies and treatment options for PD.
Danielle Mor, Ph.D., Medical College of Georgia at Augusta University
 Dr. Danielle Mor
Dr. Danielle Mor (Photo Provided)
One of the hallmarks of Parkinson's disease (PD) is the formation of Lewy bodies, which are abnormal deposits of the protein a-synuclein in the brain. However, the role of a-synuclein in PD remains unclear. Recent research suggests that a-synuclein may first appear in the neurons of the digestive tract before spreading to the brain. As part of her research into the efficacy of early interventions in the treatment of PD, Dr. Danielle Mor seeks to apply her Early-Investigator Research Award to test this "gut-to-brain" hypothesis using the nematode worm Caenorhabditis elegans (C. elegans) to understand how a-synuclein affects learning and memory, identify how gut-derived a-synuclein enters neurons, and develop novel treatments more quickly than can be accomplished using more traditional rodent models.
For more information on Dr. Mor's research, visit http://www.themorlab.org and https://www.augusta.edu/mcg/dnrm/faculty/daniellemorlab.php.
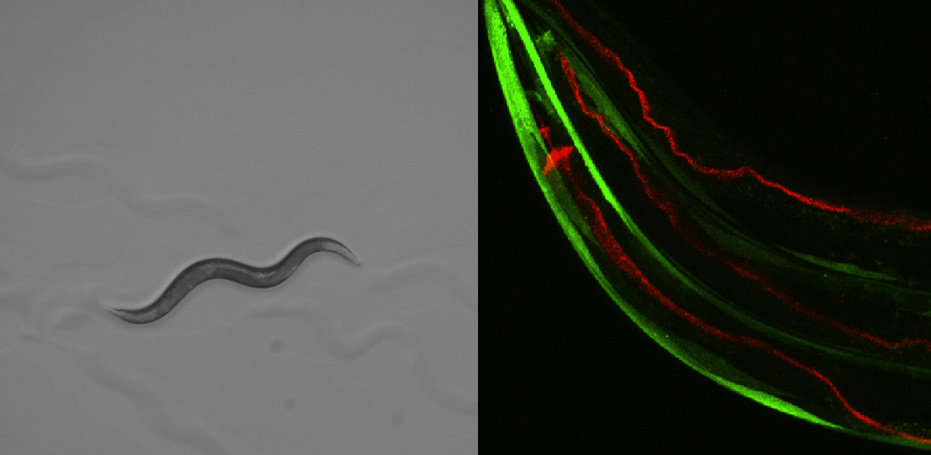 Figure 3 (left): C. elegans nematode worm. Figure 4 (right): New C. elegans models of Parkinson's disease developed by the Mor lab.
In red, the neurotoxin a-synuclein is fed to worms and can be seen in the intestinal tract (three worms are side by side in this image). Gut-derived a-synuclein then spreads to other tissues of the body and promotes disease. In green, a-synuclein expression in body wall muscle can be seen outlining the worms. Reference: Chen et al., Life Sci Alliance 2022. (Figure Provided)
Figure 3 (left): C. elegans nematode worm. Figure 4 (right): New C. elegans models of Parkinson's disease developed by the Mor lab.
In red, the neurotoxin a-synuclein is fed to worms and can be seen in the intestinal tract (three worms are side by side in this image). Gut-derived a-synuclein then spreads to other tissues of the body and promotes disease. In green, a-synuclein expression in body wall muscle can be seen outlining the worms. Reference: Chen et al., Life Sci Alliance 2022. (Figure Provided)
Giulietta M. Riboldi, M.D., Ph.D., New York University Grossman School of Medicine
 Dr. Giulietta M. Riboldi
Dr. Giulietta M. Riboldi (Photo Provided)
Early identification of Parkinson's disease (PD) is crucial for successful disease management. However, researchers have yet to fully understand the mechanisms responsible for the development of PD as well as particular biological characteristics, called biomarkers, that would allow early detection of ongoing disease processes. With support from an Early-Investigator Research Award, Dr. Giulietta Riboldi seeks to study how environmental exposures impact the behavior of immune cells by affecting the way their genes work in a cohort of people with certain non-motor clinical presentations (such as impaired sleep cycles and loss of sense of smell) or who carry specific PD-associated gene mutations that indicate an increased risk of developing PD. By correlating environmental exposures with genetic changes in the immune system, Dr. Riboldi hopes to better understand the role the immune system plays in the development of PD and identify biomarkers that clinicians can use to identify the presence of the disease earlier and target suitable preventive interventions.
Link: Epigenetics and PD
Last updated Wednesday, September 17, 2025
 An official website of the United States government
An official website of the United States government
 ) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
