Peer Reviewed Orthopaedic



Posted November 8, 2022
D. Kacy Cullen, Ph.D., and Suradip Das, Ph.D.
Corporal Michael J. Crescenz Veterans Affairs Medical Center and the University of Pennsylvania
 D. Kacy Cullen, Ph.D., and Suradip Das, Ph.D.,
D. Kacy Cullen, Ph.D., and Suradip Das, Ph.D.,
Corporal Michael J. Crescenz Veterans Affairs Medical Center and the University of Pennsylvania (Photo provided)
Summary: Volumetric muscle loss (VML) can be caused by trauma or surgery. It is defined as the loss of skeletal muscle that leads to significant loss of function. Current clinical procedures, including the use of muscle grafts and tissue-engineered muscle products, have limited success because they do not have an appropriate nerve supply, which is important for many muscle functions. Dr. D. Kacy Cullen and Dr. Suradip Das at the Corporal Michael J. Crescenz Department of Veterans Affairs Medical Center have developed a tissue-engineered product that alleviates these challenges by incorporating nerves into the product to help regenerate the surrounding muscle. The researchers recently published compelling data with evidence to suggest their tissue-engineered product leads to increased muscle growth and functional recovery in a clinically relevant VML animal model. With further development, this technology may one day help regenerate functional muscle in persons impacted by orthopaedic injury and/or VML.
Technical Details: VML is defined as loss of skeletal muscle tissue that results in substantial functional deficits in joint range of motion and skeletal muscle strength, which may cause lifelong dysfunction and disability. It can be the result of orthopaedic trauma or surgical interventions.1 The current gold standard of treatment for VML is free functional muscle transfer, but its success is limited due to donor-site morbidity, extensive operation time, and the necessity for the muscles to remain without nerve attachments for a prolonged period of time. Tissue-engineered muscle (TEM) constructs offer another treatment option but are still in their early stages of investigation. Although promising, these constructs lack a nerve supply (innervation), which is a serious impediment of their use. Therefore, development and evaluation of alternative approaches for treatment of VML is needed.
With funding from a Fiscal Year 2018 Peer Reviewed Orthopaedic Research Program Applied Research Award, Dr. Cullen and his team are using their novel mechanical bioreactors to facilitate axonal stretch growth and develop pre-innervated tissue-engineered muscle (INTEM) that can be used to promote functional re-innervation and muscle regeneration following VML. INTEMs are cultures of muscle cells or myocytes that are grown in combination with spinal motor neurons, which are responsible for carrying signals from the nervous system to the muscles, thereby inducing movement. The study team hypothesized that the presence of neurons during INTEM creation would lead to more robust cellular growth during initial fabrication, facilitate the integration of INTEMs with the host nervous system, and lead to greater functional muscle recovery in VML animal models.
For this study, Drs. Cullen and Das developed an INTEM technology that can be implanted into the muscle to promote functional muscle growth (Figure 1). While culturing the cells that would be used in the construct, the study team noted that, after 7 days of incubation, the neuron-myocyte co-culture had longer and thinner myofibers with a higher myocyte fusion index compared to the non-innervated cultures. After characterizing the INTEM constructs, integration and cell survival were assessed in a rat model with a VML injury. The VML injury was evaluated prior to implantation of the INTEM construct using non-invasive ultrasound to measure the muscle in the rat model. There was significantly more muscle volume recovery at 3 weeks with the innervated constructs compared to those with the non-innervated constructs, indicating improved cell survival with the INTEM technology.
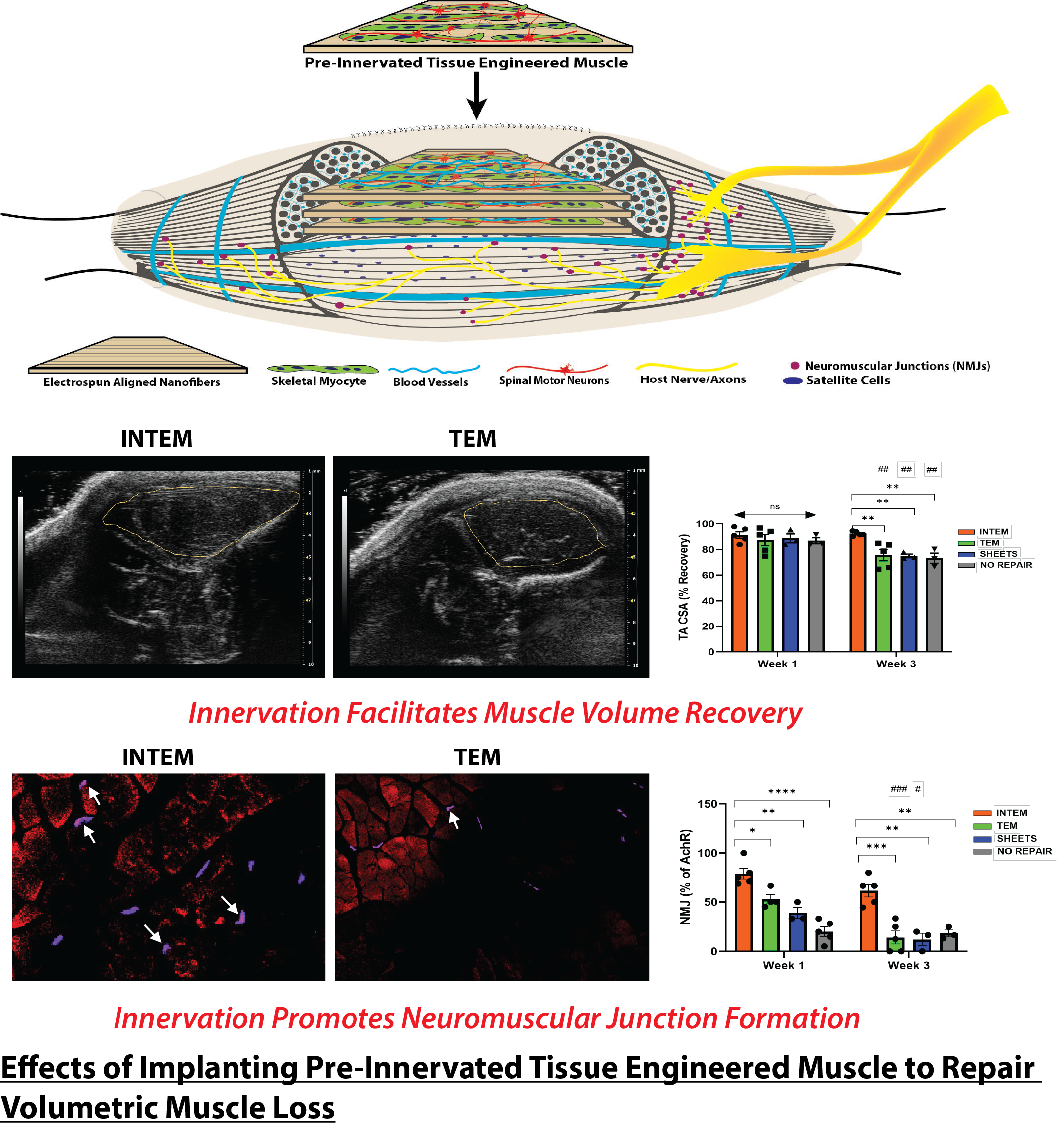
Figure 1. Innervated Tissue Engineered Muscle (INTEMs) Preserve a Pro-Regenerative Environment in Host Muscle Following VML. INTEMs comprised of coculture of rat primary motor neurons and mouse skeletal myocytes cultured on nanofiber sheets were implanted in a rodent VML model. INTEMs demonstrated higher percentage of Tibialis Anterior (TA) volume recovery and higher percentage of innervated of Acetylcholine receptors (AchRs) (indicated by colabelling of Red Synaptophysin and Purple Bungarotoxin) as compared with animals receiving non-innervated Tissue Engineered Muscle (TEM) or acellular sheets as implants as well as the no-repair control group. (Figure provided)
Three other important parameters were also investigated in the VML rat model to assess the integration of the nerve-muscle constructs: satellite cell migration, revascularization, and quantification of neuromuscular junctions (NMJs). Satellite cells have been found to be an important contributor to muscle regeneration. The study team observed a significantly higher number of satellite cells near the injury site for the INTEM group than the non-innervated group, at both 1 and 3 weeks post-repair. Revascularization of the injured area is necessary for survival of the newly implanted cells. After 3 weeks, the INTEM group had more microvessel-like structures at the injury site, indicating the potential for the constructs to form these important vascular connections. The loss of NMJs has been previously associated with neuromuscular degradation, while their presence is indicative of muscle health. At both the 1- and 3-week time points, the INTEM group had higher percentages of mature NMJs.
Taken together, the team's findings suggest the potential for pre-innervated muscle cells to significantly increase the functional regeneration of muscle at the site of injury for VML patients. In the future, Dr. Cullen and his team plan to evaluate the efficacy of these nerve-muscle constructs over extended time periods in the VML rat model, to better understand the long-term outcomes of the INTEMs on functional muscle recovery, as well as the best time point for intervention. A mature pre-innervation technology may have broad application in the functional regeneration of skeletal muscle tissue as well as cardiac and smooth muscle tissue. The ability to regenerate lost or damaged tissue could dramatically improve clinical outcomes and long-term recovery of Service Members, Veterans, and civilians impacted by traumatic injury.
Publications:
Burrell JC, Bhatnagar D, Cullen DK, et al. 2021. Tyrosine-derived polycarbonate nerve guidance tubes elicit proregenerative extracellular matrix deposition when used to bridge segmental nerve defects in swine. Journal of Biomedical Materials Research Part A 109(7):1183-1195.
Katiyar KS, Burrell JC, and Cullen DK, et al. 2021. Biomanufacturing of axon-based tissue engineered nerve grafts using porcine GalSafe Neurons. Tissue Engineering Part A 27(19-20):1305-1320.
Shultz R B, Katiyar KS, and Cullen DK, et al. 2021. Biopreservation of living tissue engineered nerve grafts. Journal of Tissue Engineering 12.
Zhang Q, Nguyen P, Le AD, et al. 2021. Harnessing 3D collagen hydrogel-directed conversion of human GMSCs into SCP-like cells to generate functionalized nerve conduits. Nature Portfolio Journal Regenerative Medicine 6:59.
Das S, Browne KD, Cullen DK, et al. 2020. Pre-innervated tissue-engineered muscle promotes a pro-regenerative microenvironment following volumetric muscle loss. Communications Biology 3:330.
Maggiore JC, Burrell JC, and Cullen DK, et al. 2020. Tissue-engineered axon-based "living scaffolds" promote survival of spinal cord motor neurons following peripheral nerve repair. Journal of Tissue Engineering and Regenerative Medicine 14(12):1892-1907.
References:
1Grogan BF and Hsu JR. 2011. Volumetric muscle loss. Journal of the American Academy of Orthopaedic Surgeons 19(1):S35-S37.
Last updated Friday, December 19, 2025
 An official website of the United States government
An official website of the United States government
 ) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
