Peer Reviewed Orthopaedic
Experimental PEG Fusion Medical Device for Immediate Physiological Recovery After Peripheral Nerve Injury



Posted July 12, 2017
Wesley Thayer, M.D., Ph.D., Vanderbilt University Medical Center, Nashville, Tennessee
Curt Deister, Ph.D., AxoGen Inc., Alachua, Florida


The use of tourniquets and body armor in modern warfare has led to an increase in the number of warfighters with survivable injuries. These complex injuries can involve multiple bones, joints, and nerves as well as various surrounding soft tissues. Peripheral nerve injury has a particularly devastating impact on military and veteran populations; it is often a major limiting factor in their functional recovery and is associated with increased morbidity. Peripheral nerve injuries also represent one of the largest source of long-term disability in Service members returning from deployment.
Currently, there are limited options for peripheral nerve repair. Nerve regeneration is a very slow process, and, even with the most advanced nerve repair techniques, the period between treatment and functional recovery is often months to years. The more severe the nerve injury, the slower the recovery will be, and, in some instances, such as when regenerating nerve fibers do not follow the correct path and "miss" their connection, no functional recovery will take place. In addition, symptoms of nerve damage, such as muscular problems, numbness, tingling, pain, and loss of sensation, can persist for years after the injury. Traumatic injury to nerves, as is commonly sustained in combat, often results in the most severe type of nerve injury, with minimal spontaneous recovery expected and surgical intervention routinely recommended. As a result, there is a critical need to develop superior treatment options for peripheral nerve injury in order to optimize recovery and restore function in Service members following orthopaedic trauma. Drs. Wesley Thayer and Curt Deister were awarded a Fiscal Year 2012 (FY12) Peer Reviewed Orthopaedic Research Program (PRORP) Translational Research Partnership Award to develop a device that promotes nerve repair and to prepare this technology for human application.
Dr. Thayer and Dr. Deister have developed several prototype devices that facilitate and standardize the delivery of polyethylene glycol (PEG) to repair and promote nerve regeneration. PEG-based nerve repair, often termed "PEG fusion," began to receive great attention in the research community after its ability to rapidly restore nerve function was demonstrated in animal models. PEG is already approved by the US Food and Drug Administration (FDA) for its use in humans, is not known to cause any major side effects, and is readily available, all of which support the translatability of PEG fusion into clinical practice.
In animal models, the current standard for PEG application is to administer the PEG solution manually through a syringe to the site where the physician has joined the injured nerve with sutures (neurorrhaphy). This method may cause small movements of the needle, resulting in sub-optimal delivery of the PEG solution or disturbance of the neurorrhaphy. Drs. Thayer and Deister developed the PEG Fusion Wrap Assist Device, which secures a short needle at the site of nerve injury and can be removed without disturbing the neurorrhaphy. They have determined that securing the needle will eliminate some of the variability caused by manual application of PEG. Furthermore, this prototype utilizes a blunt-tipped needle with a fixed penetration depth to further reduce manual variability and prevent additional nerve damage. The needle is secured to the nerve with a polymer strip, which is then wrapped around the nerve and secured to itself with a small surgical clamp. The entire device is then removed after PEG is administered. As expected, the animals that were treated using this removable device had a better functional recovery than those undergoing the current standard for PEG application.
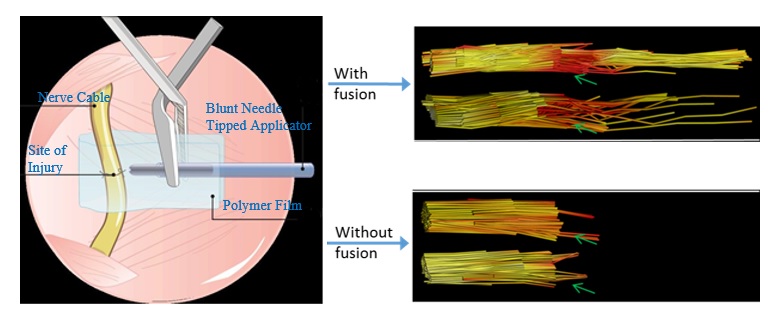
After further experimental refinement, the research team produced another prototype by modifying a clinically available implantable nerve conduit made from porcine small intestine submucosa. Drs. Thayer and Deister devised a method to more precisely align the nerve ends in a conduit using a conduit pre-loaded with "pulley" sutures. This alignment of the nerve ends is believed to be one of the physical limiters of effective PEG fusion. In addition they added a "cinching" suture to adjust the fit and improve the contact between the two faces of the severed nerve. The researchers also observed some fusion in nerves that were not treated with PEG using this second device in their animal model, indicating that further optimization of the surgical technique itself could improve functional recovery after treatment as well. Drs. Thayer and Deister are hopeful that their research will help to standardize PEG application and contribute to improvements in nerve injury repair. The novel PEG fusion delivery devices developed by Drs. Thayer and Deister bring the orthopaedic trauma field closer to restoring nerve function, which may greatly improve the quality of life of civilians and warfighters suffering from nerve injuries.
Publications:
Bamba R, Riley DC, Kelm ND, et al. 2016. A novel technique using hydrophilic polymers to promote axonal fusion. Neural Regen Res 11(4):525-528.
Bamba R, Waitayawinyu T, Nookala R, et al. 2016. A novel therapy to promote axonal fusion in human digital nerves. J Trauma Acute Care Surg 81(5):S177-S183.
Boyer RB, Kelm ND, and Riley DC. 2015. 4.7-T diffusion tensor imaging of acute traumatic peripheral nerve injury. Neurosurg Focus 39(3):E9.
Boyer RB, Sexton KW, Rodriguez-Feo CL, et al. 2015. Adjuvant neurotrophic factors with CSPG-reduced acellular nerve grafts. J Surg Res 193(2):969-977.
Riley DC, Bittner GD, Mikesh MA, et al. 2015. PEG-fused allografts produce rapid behavioral recovery after ablating sciatic nerve segments. J Neurosci Res 93(4):572-583.
Links:
Last updated Wednesday, March 12, 2025














