Peer Reviewed Medical
Clinical Development of a Novel Lung and Windpipe Sealant



Posted May 4, 2023
Daniel Weiss, M.D., Ph.D., University of Vermont and State Agricultural College

Dr. Daniel Weiss
(Photo Provided)
Lung injury of any kind, because of infection, cancer, or trauma, can result in chronic leakage of either air or fluid into or out of the lung. As current treatment options for lung leakage (e.g., chest tube drainage, surgical interventions) may lead to additional complications, new approaches are needed to improve healing and reduce negative consequences associated with lung leakage for both civilians and military Service Members. The Peer Reviewed Medical Research Program (PRMRP) has supported research targeting lung injury and/or respiratory health overall since fiscal year 2014 (FY14).
Dr. Daniel Weiss and his team had previously developed a method for generating hydrogels (a flexible, water-compatible material used to make modern contact lenses) from alginate (a protein naturally derived from seaweed). While other alginate-derived products are under development for biomedical uses, Dr. Weiss hypothesized their gel would provide the necessary elasticity, strength, and durability for use in repairing lung leakages. Thus, with an FY14 PRMRP Discovery Award to the University of Vermont, the Weiss team created and tested several modified alginate gels for use as a pleural, or lung, sealant.
Dr. Weiss identified two promising candidates for use as sealants: alginate linked to dopamine (ALG-MA-DA) and gelatin linked to dopamine (GEL-MA-DA). Both materials produced hydrogels with sufficient adhesion, strength, and elasticity for use as sealants. To test the materials' ability to withstand the air pressure generated in the lungs, his team developed new methods for burst pressure testing (i.e., the pressure at which the material fails to maintain a seal and 'burst'). They also developed application methods for the sealants: externally by aerosol spray or internally via bronchoscope. In animal studies, both sealants provided protection against air leaks for up to 1 month. Further, Dr. Weiss tested the sealants for use in trachea (i.e., windpipe) injury models and showed no air leaks for up to 2 weeks.
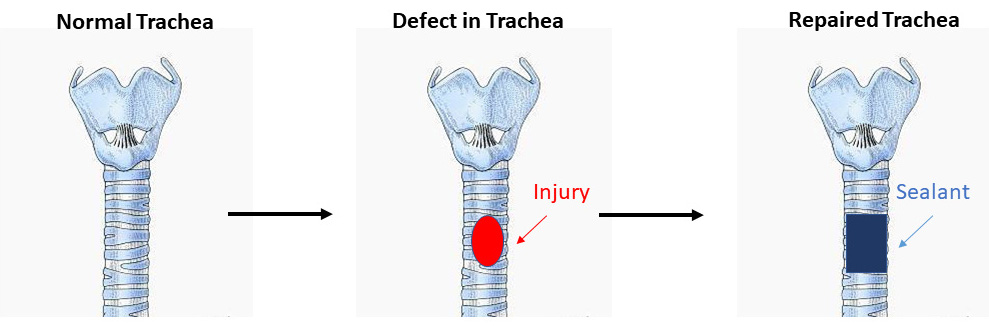
(Figure provided from the Weiss Laboratory of the University of Vermont.)
Thanks to their initial successes, Dr. Weiss continued this work through an FY18 PRMRP Expansion Award to continue to optimize their materials and to conduct longitudinal (long-term) studies of injury repair. They developed improved synthesis and manufacturing methods for both ALG-MA-DA and GEL-MA-DA, resulting in quality, "ready-to-go" patches of both materials with optimized sterilization and storage. Longitudinal studies in a small animal model show 100% survival and full recovery up to 1 year post-surgery. Similar success was found in a large animal model up to 1 month after surgery. Dr. Weiss and his team, including Dr. Christine Finck, Pediatric Surgeon and Surgeon-in-Chief at Connecticut Children's Medical Center, plan to repeat these studies on a tracheal injury model as well as in human lung tissue.
Encouraged by their findings, Dr. Weiss and his team have begun communications with the U.S. Food and Drug Administration to begin planning for future clinical studies of their sealants. Findings from the animal studies suggest their sealants would provide a single-use treatment with no need for replacement, thus no additional surgeries or treatments for patients. If successful, their sealants would provide an alternative treatment option for lung or tracheal injury and prevent extended hospital stays, reduce health care costs, and prevent complications due to lung leakage.
Publications:
Gasek N, Park HE, Uriarte JJ, et al. 2021. Development of alginate and gelatin-based pleural and tracheal sealants. Acta Biomaterialia 131:225-235.
Park HE, Gasek N, Hwang J, et al. 2020. Effect of temperature on gelation and cross-linking of gelatin and methacryloyl for biomedical applications. Physics of Fluids 32(3):033102. doi: 10.1063/1.5144896
Links:
Public and Technical Abstracts: Clinical Development of a Novel Pleural and Tracheal Sealant
Public and Technical Abstracts: Development of a Novel Alginate-Based Pleural Sealant
Last updated Wednesday, September 17, 2025
 An official website of the United States government
An official website of the United States government
 ) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
