Peer Reviewed Medical
Preclinical Advancement of Novel Mechanism-of-Action Therapeutics to Combat Type 2 Diabetes in US Veterans



Posted February 21, 2023
Harshini Neelakantan, Ph.D., Ridgeline Therapeutics, LLC

Dr. Harshini Neelakantan
(Photo Provided)
Type 2 diabetes (T2D) is a chronic condition that affects the body's ability to produce and use insulin, a hormone that regulates the amount of glucose in the bloodstream. Excess glucose, also called blood sugar, can damage the body's circulatory, nervous, and immune systems. T2D disproportionately affects Veterans. The U.S. Department of Veterans Affairs estimates that nearly a quarter of the Veterans enrolled in the Veterans Health Administration have diabetes.
Obesity is a known high-risk factor for developing T2D. Over 44% of Veterans who served in Operation Enduring Freedom, Operation Iraqi Freedom, and Operation New Dawn are obese and at risk for T2D; this obesity rate is significantly higher than the obesity rate observed in similarly aged American civilians (Wischik et al, 2019). Unfortunately, traditional weight-loss interventions like diet and exercise rarely achieve effective long-term management of obesity and T2D. The Peer Reviewed Medical Research Program (PRMRP) has addressed Metabolic Disease or Diabetes as Topic Areas since fiscal year 2014 (FY14).
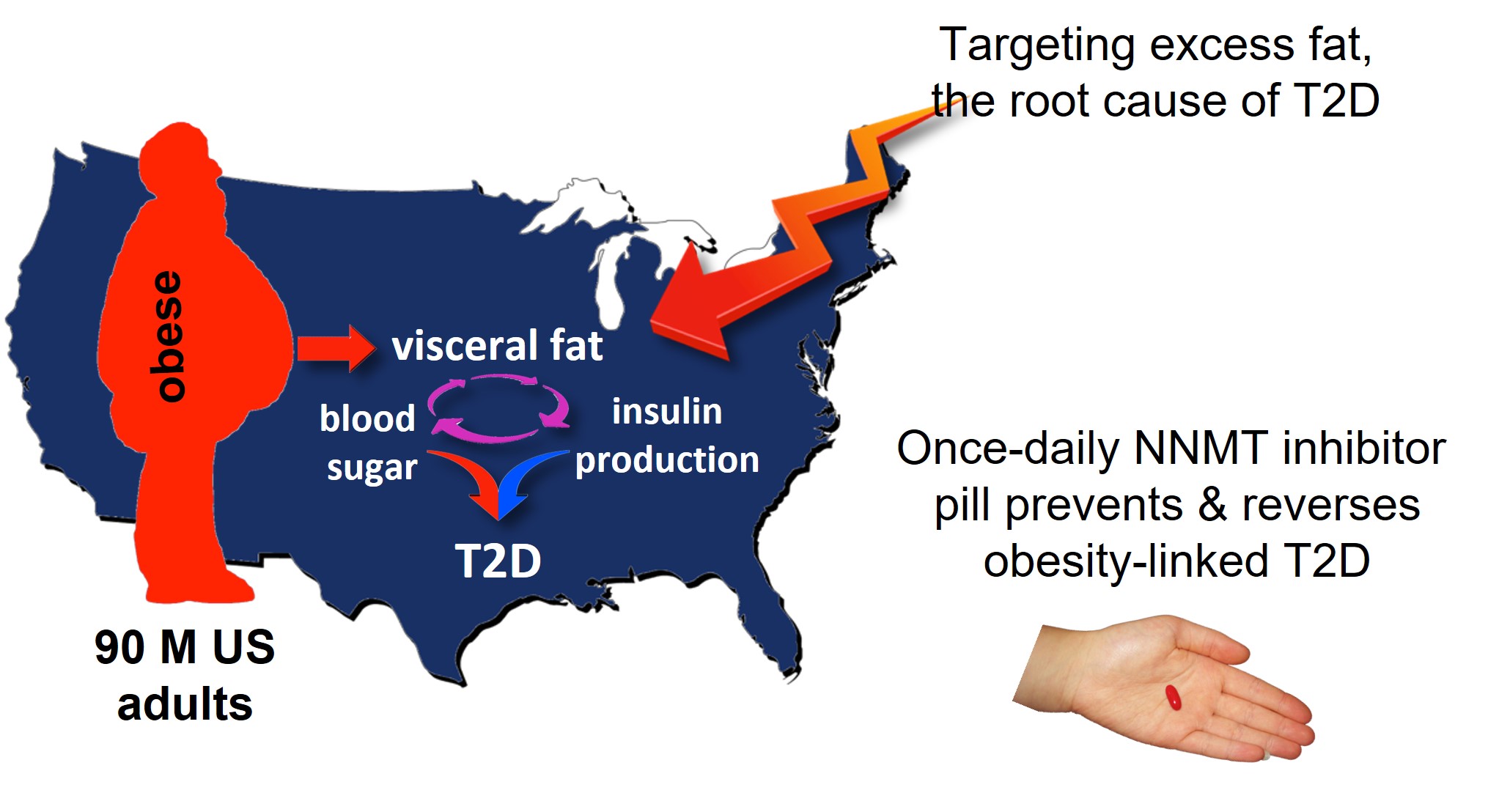
Figure 1: NNMT inhibitors target excess fat to reduce obesity and prevent or reverse obesity-induced type 2 diabetes (T2D).
With funding from an FY14 Discovery Award to the University of Texas Medical Branch at Galveston, Drs. Stanley Watowich and Harshini Neelakantan investigated an alternative approach to reducing excess body mass by identifying and successfully testing a series of first-in-class drugs that increase the energy expenditure within white adipose tissue, commonly known as body fat. These drugs decrease the activity of an enzyme called nicotinamide N-methyltransferase (NNMT), which is a key regulator of metabolism within white adipose tissue. The most promising of these drugs, an NNMT inhibitor derived from quinoline called
An FY18 Expansion Award to Ridgeline Therapeutics supported additional drug development research for
If the upcoming clinical trials prove successful, NNMT-inhibiting drugs offer a promising new approach to effectively manage body weight and reduce obesity-related diseases such as T2D among Veterans, active-duty Service Members, and the general public.
Reference:
Wischik DL, Magny-Normilus C, and Whittemore R. 2019. Risk factors of obesity in veterans of recent conflicts: Need for diabetes prevention. Current Diabetes Reports 19(9): 70. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7530827/.
Publications:
Dimet-Wiley AL, Wu Q, Wiley JT, et al. 2022. Reduced calorie diet combined with NNMT inhibition establishes a distinct microbiome in DIO mice. Science Reports 12(1):484. https://doi.org/10.1038%2Fs41598-021-03670-5.
Sampson CM, Dimet AL, Neelakantan H, et al. 2021. Combined nicotinamide N-methyltransferase inhibition and reduced-calorie diet normalizes body composition and enhances metabolic benefits in obese mice. Scientific Reports 11(1):5637. https://doi.org/10.1038%2Fs41598-021-85051-6.
Neelakantan H, Wang HY, Vance V, et al. 2017. Structure-activity relationship for small molecule inhibitors of nicotinamide N-methyltransferase. Journal of Medicinal Chemistry 60(12):5015-5028. https://pubs.acs.org/doi/10.1021/acs.jmedchem.7b00389.
Neelakantan H, Vance V, Wang HL, et al. 2017. Noncoupled fluorescent assay for direct real-time monitoring of nicotinamide N-methyltransferase activity. Biochemistry 56(6):824-832. https://doi.org/10.1021/acs.biochem.6b01215.
Neelakantan H, Vance V, Wetzel MD, et al. 2018. Selective and membrane permeable small molecule inhibitor of nicotinamide N-methyltransferase reverses diet-induced obesity in mice. Biochemical Pharmacology 147:141-152. https://doi.org/10.1016/j.bcp.2017.11.007.
Links:
Last updated Wednesday, September 17, 2025
 An official website of the United States government
An official website of the United States government
 ) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
