Peer Reviewed Medical
Development of DF-COV for the Treatment and Prevention of COVID-19 and Associated Immunopathologic Respiratory Complications



Posted March 28, 2023
Gordon Freeman, Ph.D., Dana-Farber Cancer Institute

Dr. Gordon Freeman (sitting) and colleague
SARS-CoV-2, the coronavirus responsible for COVID-19, infects a healthy cell by using projections called spike proteins to snag a protein called ACE2 on the outer membrane of a healthy cell. Once the spike protein latches onto the ACE2 receptor, it pulls the healthy cell close and drives the spike through the outer membrane, allowing the virus to release its RNA into the hijacked host. Many coronaviruses use this method to infect healthy cells. Although antibodies can target spike proteins, antibodies typically only work against a single coronavirus strain and can be rendered ineffective by mutations. As a result of these mutations, antibodies authorized by the U.S. Food and Drug Administration (FDA) to treat COVID-19 may eventually become ineffective.
In fiscal year 2020 (FY20), the Peer Reviewed Medical Research Program (PRMRP) released program announcements specifically addressing emerging viral diseases and respiratory health to counter the COVID-19 pandemic. Dr. Freeman and his team at the Dana-Farber Cancer Institute used their FY20 PRMRP Technology/Therapeutic Development Award to develop a new therapeutic, DF-COV-01, that targets spike proteins in a novel way.
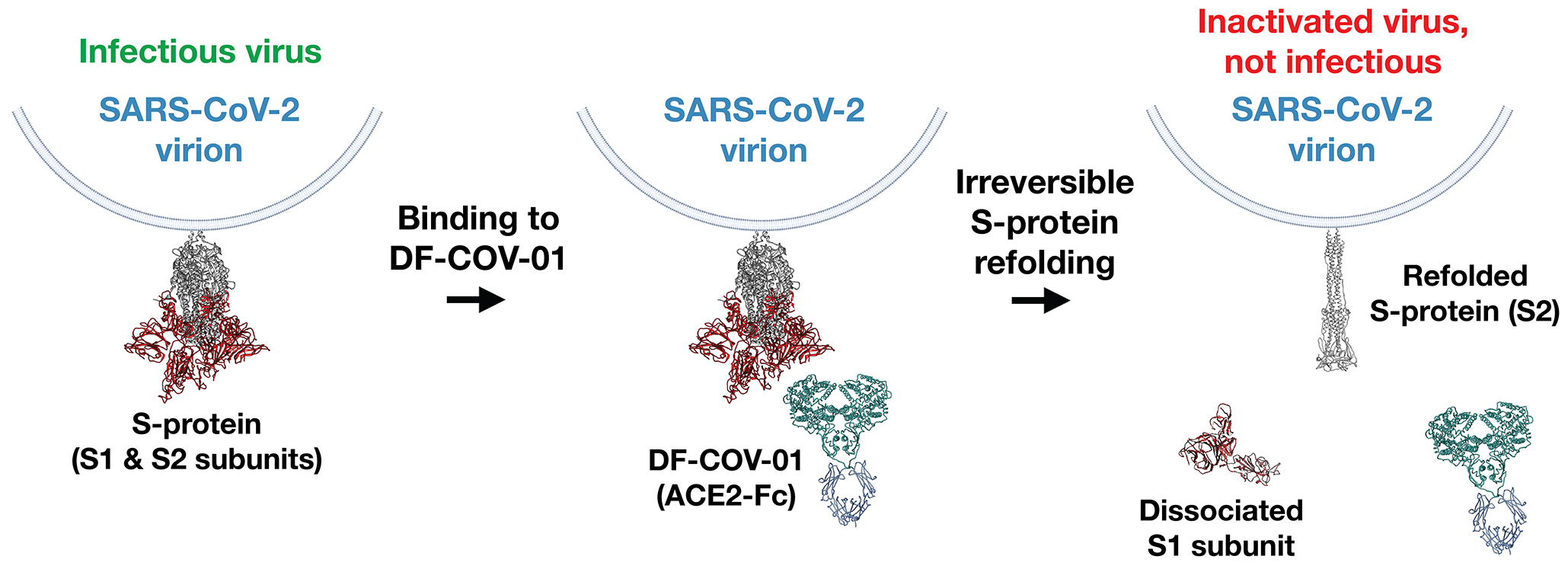 Figure 1: DF-COV-01 binds the viral S-protein and triggers its refolding, which renders the SARS-CoV-2 virus incapable of binding ACE2 and infecting human cells. (Figure Provided)
Figure 1: DF-COV-01 binds the viral S-protein and triggers its refolding, which renders the SARS-CoV-2 virus incapable of binding ACE2 and infecting human cells. (Figure Provided)
Dr. Freeman and his team began by evaluating the biological and physical characteristics of four DF-COV compounds (DF-COV-01 through DF-COV-04) to assess their ability to neutralize SARS-CoV-2 - first using infected human lung cells, and then in animal models. During their research, they discovered that changes in two structural elements in
They successfully developed a stable, high-productivity cell bank to produce DF-COV-01 and developed manufacturing and control processes in accordance with industry standards to support large-scale production of
References:
Torchia JA, Tavares AH, Carstensen LS, et al. 2022. Optimized ACE2 decoys neutralize antibody-resistant SARS-CoV-2 variants through functional receptor mimicry and treat infection in vivo. Science Advances 8(49):eabq6527. https://www.science.org/doi/10.1126/sciadv.abq6527.
Links:
Last updated Wednesday, September 17, 2025
 An official website of the United States government
An official website of the United States government
 ) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
