Peer Reviewed Medical
A Safer Alternative to Mechanical Ventilation in the Treatment of Acute Respiratory Distress Syndrome



Posted January 24, 2023
Jeffrey Borenstein, Ph.D., The Charles Stark Draper Laboratory

Dr. Jeffrey Borenstein
(Photo Provided)
Acute Respiratory Distress Syndrome (ARDS) is a medical condition in which fluid leaks into the lungs, resulting in difficulty breathing. According to Mark Siegel, M.D., at least 60 different conditions are linked to ARDS, and it remains a serious health concern for approximately 200,000 Americans diagnosed annually, notably Veterans, who are four times more likely to develop chronic lung diseases such as pulmonary fibrosis. Currently, the gold standard of treatment for ARDS is mechanical ventilation (MV), using a device which takes over breathing for patients; however, in those with highly compromised lungs, this treatment is not appropriate and could lead to additional damage. Thus, Extracorporeal Membrane Oxygenation (ECMO) was developed, which is a process to remove blood from circulation, passing it through an external device that adds oxygen and removes carbon dioxide from the blood before reintroducing it back into the blood stream. While ECMO represents a partial solution, it can be problematic due to clots that form, requiring the need for anticoagulants. In addition, this device is not appropriate for use in prolonged field care settings where Service Members require stabilization.
Acute lung injury, including ARDS, was addressed as a Topic Area by the Peer Reviewed Medical Research Program (PRMRP) from 2000-2002 and 2015-2019 (FY00-FY02, FY15-FY19) to address gaps and promote research in this field.
The Charles Stark Draper Laboratory was awarded an FY18 PRMRP Technology/Therapeutic Development Award, led by Dr. Jeffrey Borenstein, to investigate the development of a simpler and safer device for the treatment of ARDS in the prolonged field care setting. Dr. Borenstein and his team of researchers sought to develop a device within the growing field of blood oxygenators (BLOx) known as microfluidic oxygenators, where gas exchange occurs through channels on a small scale consistent with human anatomy. The project aims to facilitate improved efficiency of oxygenation. Their device differs from alternative models in three main domains: design of the device, fabrication techniques, and testing outcomes.
Dr. Borenstein's team first developed a single-layer prototype BLOx that focused on reducing elements that cause turbulence in blood flow and mimicking natural circulation to maximize gas exchange efficiency. The research team expanded on their initial success by joining a single-layer BLOx into an eight-layer stack model capable of supporting increased blood flow rates for larger-scale application (Refer to Figure 1). Throughout fabrication, they focused on safety and reproducibility, including identifying a key external source for a roll-to-roll film that could be used in the gas-transfer membrane. They also created custom housing units for the devices to ensure a secure and safe method for shipping.
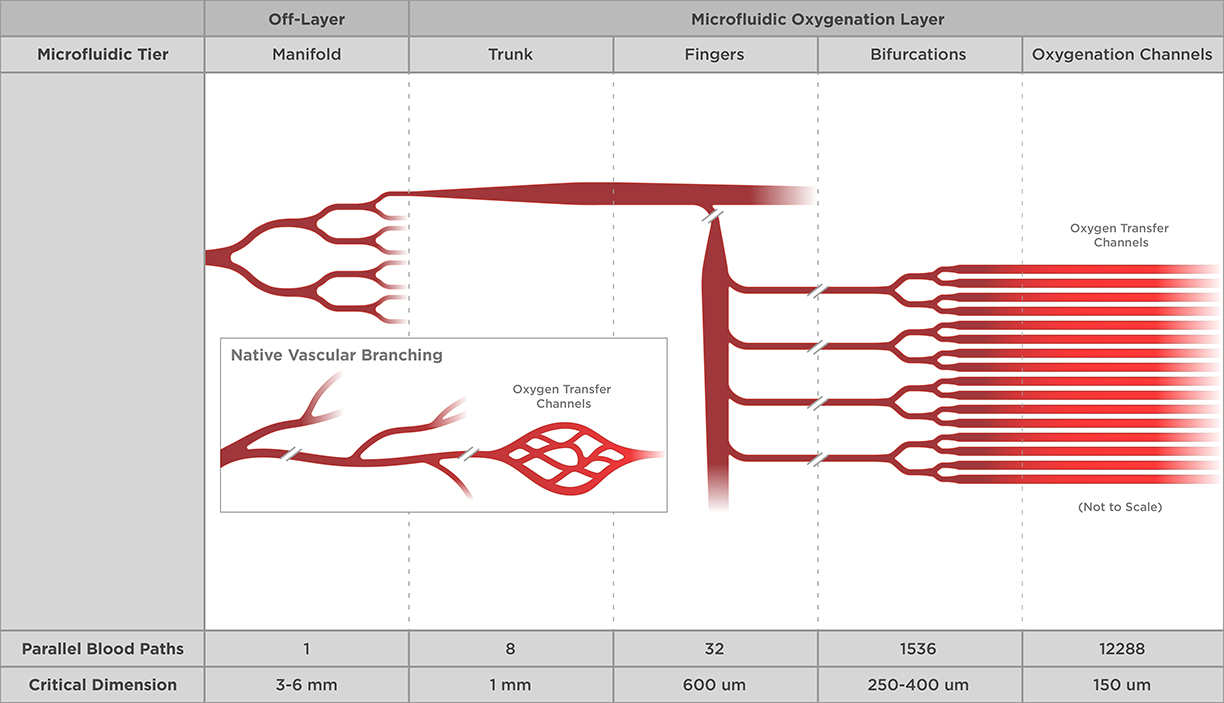
The team's attention to design detail resulted in success across three large-scale animal studies conducted at Dr. Andriy Batchinsky's Autonomous Reanimation and Evacuation Research Program in San Antonio, Texas. In these studies, blood was processed continuously through the BLOx at a rate of 750 mL/minute over a 24-hour period, an important threshold indicating readiness to move toward larger-scale studies tested over a longer duration. In each study, blood flow rate was shown to be stable, with uniform filling of each layer. Additionally, the eight-layer system demonstrated no significant increase in blood pressure levels that are typical in clot formation, noted to be a problem with conventional microfluidic oxygenator devices. Finally, their device demonstrated oxygen saturation levels that exceeded predictions in all three animal studies.
These findings show promising potential in the development of BLOx devices that can reduce reliance on anticoagulant therapy while maintaining simplicity and efficiency.
References:
Siegel M. 2022. Acute respiratory distress syndrome: Epidemiology, pathophysiology, pathology, and etiology in adults. UpToDate. Acute respiratory distress syndrome: Epidemiology, pathophysiology, pathology, and etiology in adults.
Publications:
Astor TL and Borenstein JT. 2022. The microfluidic artificial lung: Mimicking nature's blood path design to solve the biocompatibility paradox. Artificial Organs 46(7):1227-1239.
Vedula E, Isenberg B, Santos J, et al. 2022. Multilayer scaling of a biomimetic microfluidic oxygenator. ASAIO Journal 68(10):1312-1319.
Gimbel A, Hsiao J, Kim E, Lewis D, et al. 2021. A high gas transfer efficiency microfluidic oxygenator for extracorporeal respiratory assist applications in critical care medicine. Artificial Organs 45(8):E247-E264.
Santos J, Gimbel A, Peppas A, et al. 2021. Design and construction of three-dimensional physiologically-based vascular branching networks for respiratory assist devices. Lab on a Chip 21(23):4637-4651.
Santos J, Vedula E, Lai W, et al. 2021. Toward development of a higher flow rate hemocompatible biomimetic microfluidic blood oxygenator. Micromachines (Basel) 12(8):888.
Links:
Last updated Monday, December 15, 2025
 An official website of the United States government
An official website of the United States government
 ) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
