Peer Reviewed Medical
Improving Function Through Nonopioid Pain Relief in Service Members with Chronic Back Pain Following Trauma or Overuse Injury Using a Nonsurgical Therapy



Posted June 28, 2023
Joseph Boggs, Ph.D., SPR Therapeutics, Inc.

Dr. Joseph Boggs (right) with SPR Therapeutics' team of clinical research professionals studying the use of SPRINT PNS for chronic low back pain under an FY17 PRMRP Clinical Trial Award. (Photo Provided)
Low back pain (LBP) is one of the most prevalent musculoskeletal disorders among adults, with up to 84% of adults reporting LBP in their lifetime.1 LBP can be due to illness or disease, but it is commonly the result of mechanical or structural problems in the spine, discs, or muscles as a result of overuse or injury.2 If symptoms persist longer than 12 weeks, it is considered chronic LBP. Chronic, severe LBP poses a significant problem for U.S. Service Members and Veterans, with "other back problems" being the highest medical encounter category for active-duty Service Members since 2011, and Veterans are more likely than non-Veterans to report severe and chronic LBP.3 Treatments for LBP range from nonsurgical approaches, such as yoga, massage therapy, and acupuncture, to medications such as anti-inflammatory and opioid medications, and, in severe cases, surgical intervention. However, many treatments are not suitable for certain patients and others pose serious risk with no guarantee they will relieve LBP. The Peer Reviewed Medical Research Program (PRMRP) solicited musculoskeletal disorders research from fiscal year 2017 (FY17) to FY19.
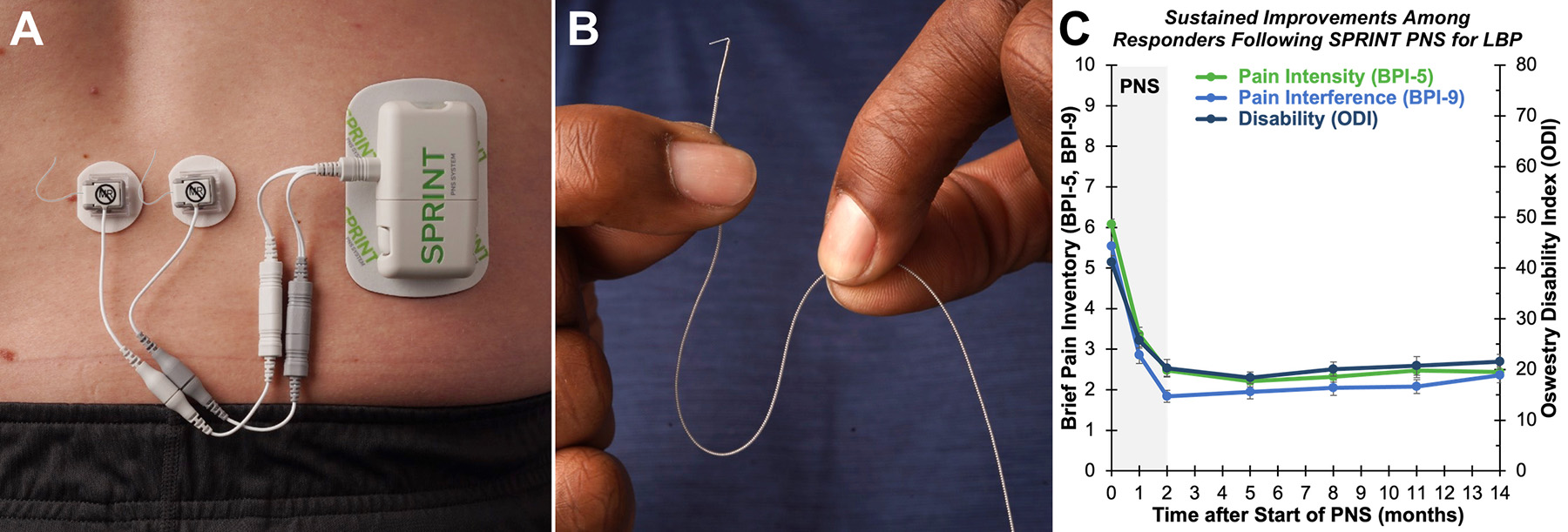
The SPRINT PNS System is a temporary (60-day) minimally invasive neurostimulation device (A) that uses a flexible, fine-wire lead (B) and has been shown to provide clinically significant improvements in pain and functional outcomes among patients with chronic LBP4 (C). The ongoing FY17 PRMRP project is expected to compare outcomes with SPRINT PNS to conventional techniques for the management of chronic, disabling LBP. (Figure Provided)
With an FY17 PRMRP Clinical Trial Award (CTA), Dr. Joseph Boggs and his team are executing a phase 2 randomized controlled trial comparing SPRINT Peripheral Nerve Stimulation (PNS) to conventional LBP management techniques. SPRINT PNS is a minimally invasive device that is designed to safely deliver electrical stimulation around the painful nerve to provide drug-free pain management. The goals of this project are to determine if the SPRINT PNS device will reduce LBP and the need for pain-relieving drugs. In addition, the study will determine if SPRINT PNS improves function in individuals with chronic LBP resulting from trauma or overuse injury. The study is finalizing participant and clinical site enrollment and expects to complete the trial enrollment and analyze the primary endpoint results in mid- to-late 2024.
The clinical validation of SPRINT PNS was supported through FY11 Peer Reviewed Orthopaedic Research Program (PRORP) and FY16 Joint Warfighter Medical Research Program (JWMRP) awards. The FY11 PRORP phase 2 clinical trial determined that PNS in a post-amputation lower limb population reduced pain in 80% of participants and decreased opioid use up to 71%. With the FY16 JWMRP award, Dr. Boggs and his team conducted a phase 3 clinical trial to test the efficacy of SPRINT PNS to relieve
The culmination of the work Dr. Boggs and his team are doing is expected to determine that PNS is a feasible pain management system, particularly for LBP and postoperative pain. Restoring musculoskeletal function and reducing the risk of dependency on pain-relieving medications, such as opioids, has high potential to improve the quality of life for Service Members, Veterans, and the public, and the capacity to help patients return to duty or work.
References:
1Chou R, Deyo R, Friedly J, et al. 2016. Noninvasive treatments for low back pain. Comparative Effectiveness Reviews, No. 169. Rockville, MD: Agency for Healthcare Research and Quality. https://www.ncbi.nlm.nih.gov/books/NBK350262
2National Institute of Arthritis and Musculoskeletal and Skin Diseases. Back Pain Basics. https://www.niams.nih.gov/health-topics/back-pain
3VA/DoD Clinical Practice Guideline. 2022. The diagnosis and treatment of low back pain. Washington, DC: U.S. Government Printing Office. https://www.healthquality.va.gov/guidelines/Pain/lbp/VADoDLBPCPGFinal508.pdf
4Gilmore CA, Deer TR, Desai MJ, et al. 2023. Durable patient-reported outcomes following 60-day percutaneous peripheral nerve stimulation (PNS) of the medial branch nerves. Interventional Pain Medicine 2(1):100243. https://doi.org/10.1016/j.inpm.2023.100243
Links:
Last updated Wednesday, September 17, 2025
 An official website of the United States government
An official website of the United States government
 ) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
