Peer Reviewed Medical
Development of an Innovative Combination Therapy Against Multidrug-Resistant Bacteria




Posted November 30, 2022
Evgeny Nudler, Ph.D., and Alexander Serganov, Ph.D., New York University
 Dr. Evgeny Nudler
Dr. Evgeny Nudler
(Photo provided)
 Dr. Alexander Serganov
Dr. Alexander Serganov
(Photo provided)
Antibacterial resistance, or the ability of bacteria to resist the effects of drugs designed to kill them, is an urgent public health crisis according to the Centers for Disease Control and Prevention. This resistance increases the risk that an infection will spread and can lead to serious illness, including death. Further, some bacteria, known as multiple drug resistant organisms (MDROs), have developed resistance to many available drugs, making infections more difficult to treat. Antimicrobial resistance was addressed by the Peer Reviewed Medical Research Program (PRMRP) during fiscal years 2016-2019 (FY16-FY19).
In FY17, Drs. Evgeny Nudler and Alexander Serganov each were supported by a PRMRP Investigator-Initiated Research Award - Partnering PI (Principal Investigator) Option to develop a conceptually novel therapy to treat MDROs and manage combat-related drug-resistant infections. Rather than simply identifying new antibiotics, their goal was to find a strategy to potentiate currently available antibiotics. According to a 2014 Proceedings of the National Academy of Sciences article by Dwyer et al., many antibiotics induce oxidative stress on bacterial cells, which contributes to drug lethality. In response, bacteria produce non-toxic amounts of hydrogen sulfide (H2S) to protect themselves from the oxidative damage and death. Drs. Nudler and Serganov focused on targeting H2S production in their quest to find a way to augment current antibiotic therapy.
Drs. Nudler and Serganov and their teams identified an enzyme called bacterial cystathionine γ-lyase (bCSE) as the primary source of H2S production in Pseudomonas aeruginosa (P. aeruginosa) and Staphylococcus aureus (S. aureus), two MDROs primarily responsible for hospital-acquired infections. This finding makes bCSE a promising drug target to sensitize these bacteria to current therapies. The Nudler and Serganov teams utilized the molecular structure of bCSE to design novel compounds that inhibit bCSE function. They successfully designed three compounds capable of significantly inhibiting H2S production in S. aureus and P. aeruginosa. The inhibitors suppressed antioxidation defenses and the ability of bacteria to employ antibiotic resistance and tolerance mechanisms, such as biofilms and persistence.
Using antibiotic-resistant strains of S. aureus and P. aeruginosa, the team tested the effects of the three bCSE inhibitors compared to standard antibiotics (e.g., gentamycin) on bacterial growth. While the bCSE inhibitors and standard antibiotics individually had minimal impact on bacterial growth, in combination they abolished all bacterial growth. The team then tested the three bCSE inhibitors in mouse models of S. aureus and P. aeruginosa infections. Similar to the earlier experiments, mice were treated with either a bCSE inhibitor or an antibiotic, or a combination thereof. The combinatorial treatment led to an increase in life span for mice infected with S. aureus and a decrease in bacterial burden for mice infected with P. aeruginosa when compared to the individual treatments or controls. The results of this study demonstrate that inhibiting bCSE in drug-resistant bacteria sensitized them to current therapeutics and improved outcomes in mouse models of bacterial infection.
Drs. Nudler and Serganov's research teams' paradigm-shifting results have been published in articles in Nature Communications, 2019, Proceedings of the National Academy of Sciences, 2020, and Science, 2021. Their research demonstrates, for the first time, that combining bCSE inhibitors with clinical antibiotics can overcome the acquired multi-drug resistance and tolerance in bacteria associated with wounds and burns, and present in both field and hospital environments. This line of research could prove to be invaluable in the fight against MDROs and the public health threat they pose to Service Members, Veterans, and the general public.
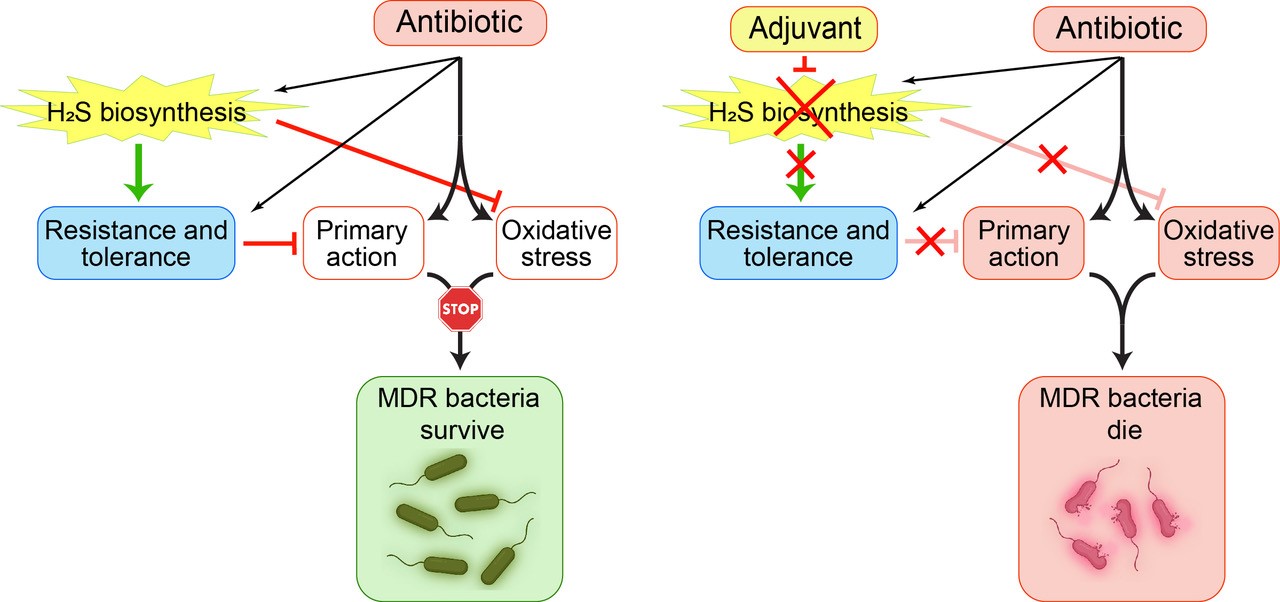
Figure 1: Describing the Relationships between H2S Biogenesis and Antibiotic Tolerance
(Left) In addition to their primary action, some antibiotics stimulate oxidative stress, which damages cell components. Bacterial cells defend themselves by producing H2S, which mitigates oxidative stress and participates in other tolerance mechanisms. As a result, bacteria can survive an antibiotic challenge by producing H2S. (Right) Specific inhibitors of H2S biogenesis reduce resistance to oxidative stress and other tolerance mechanisms, thereby improving antibiotics' potency.
(Figure provided)
Publications:
Luhachack L, Rasouly A, Shamovsky I, and Nudler E. 2019. Transcription factor YcjW controls the emergency H2S production in E. coli. Nature Communications 10:2868. doi: 10.1038/s41467-019-10785-x.
Mironov A, Seregina T, Shatalin K, et. al. 2020. CydDC functions as a cytoplasmic cystine reductase to sensitize Escherichia coli to oxidative stress and amino glycosides. Proceedings of the National Academy of Sciences 117(38):23565-23570. doi: 10.1073/pnas.2007817117.
Shatalin K, Nuthanakanti A, Kaushik A, et. al. 2021. Inhibitors of bacterial H2S biogenesis targeting antibiotic resistance and tolerance. Science 372, 1169-1175. doi: 10.1126/science.abd8377.
Links:
Public and Technical Abstracts: Development of Innovative Combination Therapy Against Multidrug-Resistant Bacteria - Dr. Evgeny Nudler
Public and Technical Abstracts: Development of Innovative Combination Therapy Against Multidrug-Resistant Bacteria - Dr. Alexander Serganov
Last updated Wednesday, September 17, 2025
 An official website of the United States government
An official website of the United States government
 ) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
