Peer Reviewed Medical
Development of (R)-ND-336 for the Treatment of Diabetic Foot Ulcers



Posted August 24, 2022
Mayland Chang, Ph.D., University of Notre Dame
Diabetes affects more than 30 million individuals in the United States, 25% of whom are Veterans.1 While diabetes specifically involves dysregulation of blood sugar, common complications of the disease includes the body's inability to heal wounds, specifically Diabetic Foot Ulcers (DFUs). Current treatment options range from surgical removal of the damaged tissues to amputation of the foot. Becaplermin is the only drug approved by the U.S. Food and Drug Administration to treat DFU, but often is not prescribed due to modest efficacy. The lack of available and effective therapies for DFUs contributes to the over 100,000 lower-limb amputations needed in the United States annually.2 Further, the 5-year mortality rate for individuals who have undergone DFU surgical amputation is 30.5 percent,3 suggesting amputation is not a reliable treatment option for DFUs and reinforces the need for better alternatives.

Dr. Mayland Chang
(Photo Provided)
With the support of a Fiscal Year 2018 Peer Reviewed Medical Research Program Technology/Therapeutic Development Award, Dr. Chang and her team identified enzymes, known as matrix metalloproteinases (MMPs), are responsible for preventing DFUs from healing and targeted these enzymes through the development of a potential topical therapeutic, (R)-ND-336. Dr. Chang isolated MMPs present in patients with DFUs, specifically MMP-8 and MMP-9. These MMPs are not only responsible for normal healing, but also play a role in how DFUs develop. MMP-8 assists with wound repair, while MMP-9 specifically prevents DFUs from healing. Dr. Chang's data suggests there is a significant increase in MMP-9 levels found in severe, infected DFUs compared to initial or mildly infected DFUs, and treatment with (R)-ND-336 prevents MMP-9 from inhibiting wound healing, increasing efficiency and accelerating wound healing in animal models.
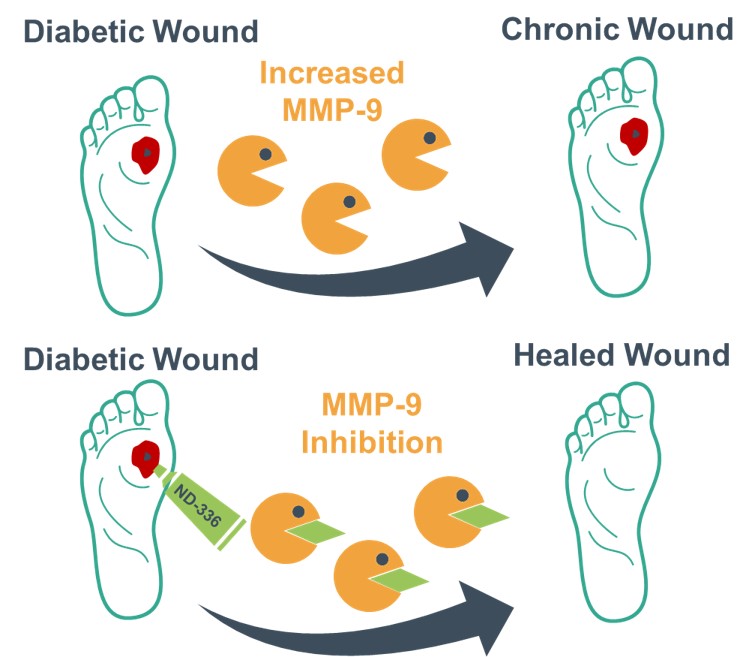
Figure 1: Increased amounts of the enzyme MMP-9 prevents DFU from healing. Topical application of (R)-ND-336 inhibits MMP-9, restoring wound healing.
(Photo Provided)
In two 2021 publications in ACS Pharmacology & Translational Science, Dr. Chang and her team compared the pharmacological properties of (R)-ND-336 to treatment with and without the antibiotic linezolid. Dr. Chang found (R)-ND-336 alone or in combination with the antibiotic accelerated healing of diabetic ulcers in infected animal models. Dr. Chang plans to advance (R)-ND-336 to human clinical trials for the treatment of DFUs. Dr. Chang is also investigating additional potential applications for (R)-ND-336 where MMPs are implicated in disease progression, including healing of pressure ulcers and treatment of benign ocular growths.
More recently, the drug successfully underwent toxicological testing, including repeated dosing in animal models, providing the necessary risk assessment for further consideration. The initiation of a Pre-Investigational New Drug Application (Pre-IND) meeting with the FDA is underway. In addition to discussing the preclinical data that would support testing (R)-ND-336 in clinical trials, Dr. Chang said, "We will discuss with the FDA whether (R)-ND-336 can receive fast track or breakthrough therapy review, which can accelerate development of (R)-ND-336." When describing plans for future clinical trials, Dr. Chang reported on a new company to support the development of (R)-ND-336. 'salvePeds is the sponsor of the IND and of clinical trials. SalvePeds is raising funds for the conduct of phase 1 clinical trials." These funds, combined with support from the PRMRP, could advance the (R)-ND-336 topical gel to become the first therapeutic treatment for DFUs that targets MMP-9. If successful, this novel approach would change the treatment paradigm for DFUs, reducing the need for amputations and decreasing the mortality rate for this common complication of diabetes.
Publications:
Peng Z, Nguyen TT, Song W, et al. 2021. The selective MMP-9 inhibitor (R)-ND-336 alone or in combination with linezolid accelerates wound healing in infected diabetic mice. ACS Pharmacology & Translational Science 4(1):107-117. https://doi.org/10.1021/acsptsci.0c00104.
Gabriel CE, Nguyen TT, Gargano EM, et al. 2021. Metabolism of the selective matrix metalloproteinase-9 inhibitor (R)-ND-336. ACS Pharmacology & Translational Science 4(3):1204-1213. https://doi.org/10.1021/acsptsci.1c00063.
Chang M and Nguyen TT. 2021. Strategy for treatment of infected diabetic foot ulcers. Accounts of Chemical Research 54(5):1080-1093. https://dx.doi.org/10.1021/acs.accounts.0c00864.
Peng Z, Nguyen TT, Wang M, et al. 2022. Proteomics identification of targets for intervention in pressure ulcers. ACS Chemical Biology 17(6):1357-1363. https://doi.org/10.1021/acschembio.2c00382.
Masitas C, Peng Z, Wang M, et al. 2022. Matrix metalloproteinase-14 as an instigator of fibrosis in human pterygium and its pharmacological intervention. ACS Pharmacology & Translational Science in press. https://doi.org/10.1021/acsptsci.2c00125.
References:
1Liu Y, Sayam S, Shao X, et al. 2017. Prevalence of and trends in diabetes among Veterans, United States, 2005-2014. Preventing Chronic Disease 14:E315. https://doi.org/10.5888/pcd14.170230.
2Centers for Disease Control and Prevention. 2020. National Diabetes Statistics Report. U.S. Department of Health and Human Services, Atlanta, Georgia. https://pmc.ncbi.nlm.nih.gov/articles/PMC8236303/.
3Armstrong DG, Swerdlow MA, Armstrong AA, et al. 2020. Five year mortality and direct costs of care for people with diabetic foot complications are comparable to cancer. Journal of Food and Ankle Research 13(1):16. https://doi.org/10.1186/s13047-020-00383-2.
Links:
Public and Technical Abstracts: (R)-ND-336 for the Treatment of Diabetic Foot Ulcers
Last updated Friday, December 19, 2025
 An official website of the United States government
An official website of the United States government
 ) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
