Peer Reviewed Medical
Novel Liver-Stage-Active Antimalarials




Posted April 22, 2022
Jane X. Kelly, Ph.D., Portland VA Medical Center
LTC Brandon Pybus, Ph.D., Walter Reed Army Institute of Research
 Dr. Jane X. Kelly
Dr. Jane X. Kelly
 Dr. Brandon Pybus
Dr. Brandon Pybus
Malaria is an infectious disease spread by mosquitos carrying Plasmodium parasites. Following a complex life cycle, these parasites first grow in the liver (liver-stage) before multiplying in red blood cells (blood-stage) to be ingested by a new mosquito.1 Only blood-stage parasites cause symptoms of malaria, increasing the difficulty of both treatment and prevention approaches for reducing malaria infections. In 2019 alone, the World Health Organization estimated 229 million malaria cases and 409,000 malaria deaths globally, with children and pregnant women at highest risk of death from infection.2 Artemisinin-based combination therapies serve as front-line treatments across the globe, but with resistance to artemisinin on the rise and resistance to other antimalarials (e.g., quinolines) prevalent, new therapeutics are needed to address malaria infections. In 2009, Dr. Jane Kelly and colleagues described a potential new class of antimalarial drugs: dual-function acridones. Their publication in Nature showed an acridone compound with novel antimalarial activity and enhanced response to quinolines, even in quinoline-resistant parasites.3 Further, these acridone compounds showed activity against both blood-stage and liver-stage parasites, and their antimalarial activities were patented in 2013.4 With this preliminary data in hand, Dr. Kelly and her collaborators at the Walter Reed Army Institute of Research (WRAIR) were named as Principal Investigators (PIs) on a Fiscal Year 2013 (FY13) Peer Reviewed Medical Research Program (PRMRP) Investigator-Initiated Research Award - Partnering PI Option to synthesize and identify potential acrodine-based drug candidates. Between FY13 and FY17, Drs. Victor Melendez, Lisa Read, Charles Bane, Brian Vesely, and COL Mara Kreishman-Deitrick have contributed as Partnering PIs. This work continued to be supported by an FY16 PRMRP award and (then MAJ) Brandon Pybus took on the mantle of Partnering PI at WRAIR.
During the design and testing of new compounds, Dr. Kelly developed a new method for acridone synthesis involving a microwave heating technique, which was published in RSC Advances in 2019. The new method shortened the reaction time from up to 24 hours to 1-2 minutes. Meanwhile, LTC Pybus developed a high-throughput liver-stage screening assay to assess the compounds' activity for both prophylaxis (early liver-stage treatment) and radical cure (late liver-stage treatment). After generating and testing over 100 new candidates in cell models and mouse models, the top three compounds were tested against clinical isolates from Uganda, and the results were published in the Journal of Medicinal Chemistry in 2019 and 2020. Of these, T111 showed the most potent activity and, following promising preclinical safety studies, was selected as the lead compound for further development. T111 also had enhanced activity when paired with tafenoquine, which was approved for antimalarial use in the United States in 2018. These compounds show the potential for an improved and total cure of malaria as well as a lower drug dose leading to less toxic effects, which could revolutionize current treatment approaches for malaria.
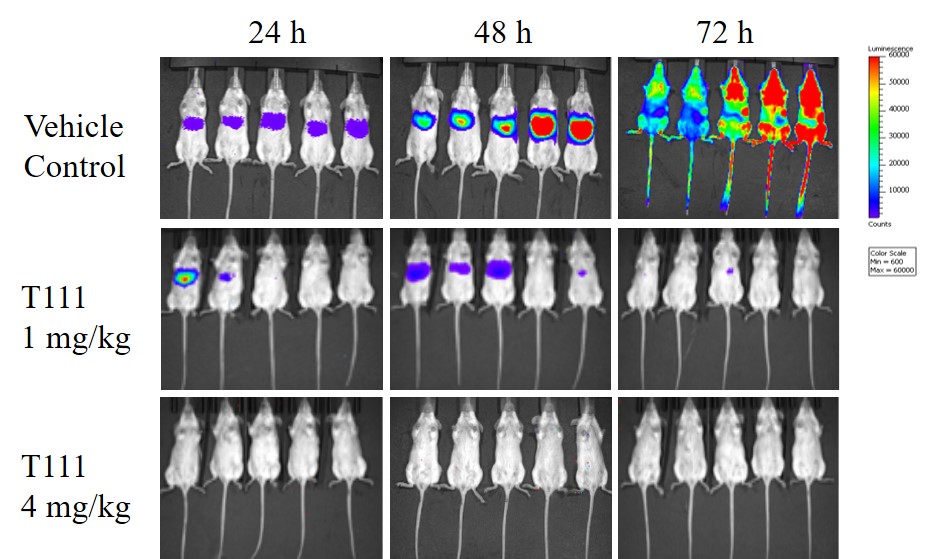
Figure 1: Bioluminescence images representing the number of parasites in mice at 24, 48 and 72 hours after an infection. Mice were treated with either the lead drug candidate, T111, at 1 mg/kg or 4 mg/kg (middle and bottom row) or a control (top row). Blue = low number of parasites, Green = medium number of parasites, Red = high number of parasites.
These research endeavors resulted in multiple publications and additional funding from the National Institutes of Health (1R01AI158533). Dr. Kelly and LTC Pybus recently received a recommendation for funding for a FY21 PRMRP Expansion Award - Funding Level 3 - Partnering PI Option to continue development of their drug candidates for eventual testing in clinical trials. Further, Dr. Kelly and LTC Pybus exemplify a productive research partnership, combining their scientific expertise as well as the resources available to them at the Portland VA Medical Center and WRAIR to make great impacts in antimalarial research. Their discoveries aim to benefit not only those who live in malaria-endemic countries but also U.S. Service Members exposed during military service and all at risk for infection.
Publications:
Kancharla P, Dodean RA, Li Y, et al. 2020. Lead optimization of second-generation acridones as broad-spectrum antimalarials. Journal of Medicinal Chemistry 63(11):6179-6202.
Dodean RA, Kancharla P, Li Y, et al. 2019. Discovery and structural optimization of acridones as broad-spectrum antimalarials. Journal of Medicinal Chemistry 62(7):3475-3502.
Kancharla P, Dodean RA, Li Y, and Kelly JX. 2019. Boron trifluoride etherate promoted microwave-assisted synthesis of antimalarial acridones. RSC Advances 9:42284.
References:
1Centers for Disease Prevention and Control. About Malaria. https://www.cdc.gov/dpdx/malaria/index.html
2World Health Organization. 2020. World Malaria Report 2020. https://www.who.int/publications/i/item/9789240015791.
3Kelly JX, Smilkstein MJ, Brun R, et al. 2009. Discovery of dual function acridones as a novel antimalarial chemotype. Nature 459(7244):270-273.
4Riscoe MK, Winter R, Kelly JX, et al. 2013. Acridone compounds. US Patent 8,592,447 B2, filed November 13, 2007; issued November 26, 2013.
Links:
Public and Technical Abstracts: Novel Synergistic Antimalarials with Gametocytocidal Activity
Public and Technical Abstracts: Novel Liver-Stage-Active Antimalarials
Public and Technical Abstracts: Preclinical Development of Novel Liver Stage Active Antimalarials with Radical Cure Potential
(Under Negotiation, Pending Award)
Press Release: NIH Grants Support the Development of Treatments for Major Diseases
Last updated Friday, December 19, 2025
 An official website of the United States government
An official website of the United States government
 ) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
