Peer Reviewed Medical
Prevention of Post-traumatic Osteoarthritis with CDK9 Inhibitors



Posted December 30, 2022
Dominik Haudenschild, Ph.D., University of California, Davis

Dr. Dominik Haudenschild
(Photo Provided)
Osteoarthritis (OA) is a painful and disabling disease caused by the breakdown of the cartilage lining between two bones within a joint. While OA may be viewed by some as an inevitable consequence of aging, arthritis continues to be one of the leading causes of disability among U.S. adults between the ages of 18-64 (Theis et al., 2018). In fact, almost all common joint injuries have been shown to progress into post-traumatic OA (PTOA) within 2-10 years after an acute injury. Joint injury leads to local inflammation that quickly damages the surrounding heathy joint tissues. Animal studies show that limiting the initial joint inflammation after an injury can prevent PTOA. However, current clinical treatments do not address early inflammation after an injury and, therefore, do not protect healthy tissues from being further damaged and prevent PTOA. Instead, treatments focus only on reducing pain and disability after OA has developed. Therefore, targeting the window of time immediately after an injury is essential to reducing the disability associated with arthritis that can result in pain, a decline in mobility, and reduced quality of life, especially for military personnel for whom joint injuries are common.
The Peer Reviewed Medical Research Program funded a Fiscal Year 2011 (FY11) Investigator-Initiated Research Award to the University of California, Davis, led by Dr. Haudenschild. During this time, Dr. Haudenschild focused on identifying the cellular and molecular drivers of OA immediately following injury. More specifically, he and his team successfully demonstrated that through the inhibition of Cdk9, a protein essential for proper control of an array of cellular functions, they could reduce early inflammatory responses that occur after injury and lead to OA. These findings exceeded their expectations, leading to a subsequent FY17 Technology/Therapeutic Development Award addressing the FY17 PRMRP Topic Area PTOA.
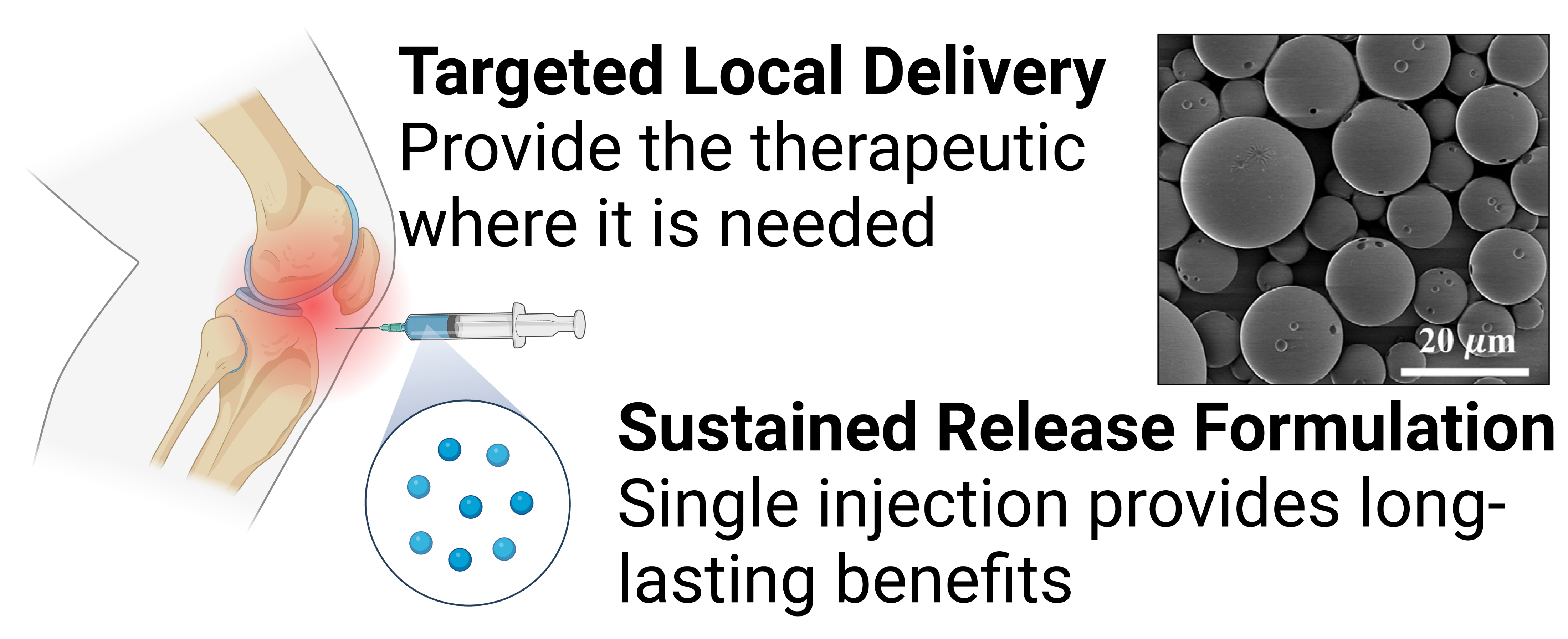 Figure Provided
Figure Provided
To further expand upon their previous findings, the team sought to improve the specific formulation of the Cdk9 inhibitor, flavopiridol, through the development of a drug-delivery vehicle that allows for slow release to offer continued joint protection. This drug-delivery vehicle recently received U.S. Food and Drug Administration (FDA) approval for an anti-inflammatory medication used for treatment of pain in individuals with OA. The second aim of this FY17 award was to assess the efficacy of the Cdk9 inhibitor in a large-animal model, in this case sheep, to move the research one step closer to application in human trials.
Despite the challenges and many obstacles due to the COVID-19 pandemic, Dr. Haudenschild and his experienced team continued to analyze the RNA sequencing data previously generated in mice treated with the Cdk9 inhibitor. Further analysis of this data helped to refine their treatment strategy and strengthen their patent position by providing more evidence and insight into how inhibition of Cdk9 can reduce the progression of OA.
They also focused on the development of a specific formulation of the drug-delivery vehicle for flavopiridol, which included developing and validating methodology to measure the amount of flavopiridol in several animal models, both locally (within the joint) and systemically (in the blood). They successfully demonstrated that flavopiridol is stable for at least 30 months when stored properly, and that the medication remained in the knee joint of a large animal model for at least 2-4 weeks after injection without adverse effects. These findings highlight the feasibility and safety, in addition to the already proven efficacy, of a potent use for flavopiridol to reduce the onset of PTOA.
Their outcomes allowed the team to obtain approval from the FDA to expedite phases of clinical trials by allowing them to bypass traditional phase 1 work in healthy human controls. This could corroborate their animal findings in humans and, therefore, validate quicker translation to clinical use for those at risk of developing PTOA. To date, they have successfully obtained exclusive license for the use of Cdk9 inhibitors and other inhibitors to reduce cartilage degradation and inhibit inflammation, totaling six patents for their commercial partner.
Dr. Haudenschild's work lays the framework for developing a commercial drug product of flavopiridol as an injectable medication that can be administered by medical professionals in doctor's offices, veterinary clinics, and on the sidelines of sporting competitions to treat joint injuries and joint OA. This work shifts the focus from the wait-and-see approach of the current model for managing joint injuries, to a more proactive treatment approach that focuses on prevention of PTOA. Early treatment after an injury to prevent the onset of secondary damage can reduce healthcare burden and, more importantly, improve the overall quality of life of individuals who would otherwise face years of disability. Successful development of this drug will have far-reaching impacts on individuals at risk of suffering from PTOA, especially Service Members who are more prone to joint injuries.
Publications:
Sangsuwan R, Yik JHN, Owen M, et al. 2022. Intra-articular injection of flavopiridol-loaded microparticles for treatment of post-traumatic osteoarthritis. Acta Biomaterialia 149:347-358. doi: 10.1016/j.actbio.2022.06.042.
Leale DM, Li L, Settles M, et al. 2022. A two-stage digestion of whole murine knee joints for single-cell RNA sequencing. Osteoarthritis and Cartilage Open. 4(4):100321.
Fukui T, Yik JHN, Doyran B, et al. 2021. Bromodomain-containing-protein-4 and cyclin-dependent-kinase-9 inhibitors interact synergistically in vitro and combined treatment reduces post-traumatic osteoarthritis severity in mice. Osteoarthritis and Cartilage 29(1):68-77. doi: 10.1016/j.joca.2020.07.012.
References:
Theis KA, Roblin D, Helmick CG, and Luo R. 2018. Prevalence and causes of work disability among working age U.S. adults, 2011-2013, NHIS. Disability and Health Journal 11(1):A1-A168. doi: 10.1016/j.dhjo.2017.04.010.
Links:
Public and Technical Abstracts: Prevention of Posttraumatic Osteoarthritis with CDK9 Inhibitors
Last updated Wednesday, September 17, 2025
 An official website of the United States government
An official website of the United States government
 ) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
