Peer Reviewed Medical
Amicidin-α Surgical Gel for Prevention of Infections in Surgery and Trauma



Posted February 2, 2022
Michael Bevilacqua, M.D., Ph.D., Amicrobe, Inc.

Dr. Michael Bevilacqua
The potential for infection is a major concern for traumatic and post-surgical wounds, including combat-related wounds. When skin tissue is compromised, its protective barrier and antimicrobial properties are lost and the risk of localized infection increases, which can lead to systemic infection and sepsis. The growing prevalence of multidrug-resistant bacteria, which are no longer controlled or killed by antibiotics, add an additional level of risk for these injuries. Researchers are developing novel antimicrobial molecules made of amino acids - the building blocks for peptides and proteins - as a means to prevent and treat trauma- or surgery-induced infections. These synthetic peptides/proteins contain particular arrangements of amino acids that display antimicrobial activity as well as other desirable properties that make them suitable for various healthcare applications.
Amicidin-α Surgical Gel, previously known as A-Blocks Surgical Hydrogel, is one such product being developed by Amicrobe, Inc. This antimicrobial therapeutic gel consists of Amicidin-α, a synthetic protein antimicrobial, which has the potential to reduce the occurrence of infection in traumatic and post-surgical wounds. In a 2017 Macromolecular Bioscience publication, Dr. Michael Bevilacqua and his team demonstrated the broad effects of synthetic protein-based antimicrobials on many species of bacteria, including multidrug-resistant strains, as well as yeast. In addition to their antimicrobial properties, the team reported that the Amicidin-α-like synthetic proteins formed hydrogels that could act as a temporary barrier for exposed tissues. These hydrogels are three-dimensional structures that can swell and hold water, enabling them to form protective barriers against microbes on the skin. They also have the ability to spread easily and fill small spaces in wounds and thus may become an excellent treatment option because they increase protection from infection while supporting a wound healing environment.
With support from a Fiscal Year 2015 Peer Reviewed Medical Research Program Technology/Therapeutic Development Award to Amicrobe, Inc., Dr. Bevilacqua and his team conducted studies to optimize the manufacture and sterilization of Amicidin-α Surgical Gel. These late-stage preclinical steps included the necessary nonclinical safety studies that will allow the product developers to prepare for clinical trials. Multiple batches of Amicidin-α Surgical Gel were produced and studied to determine its consistency and quality and to help ensure its safety. After meeting Good Manufacturing Practices standards to produce a batch for use in initial nonclinical safety studies, Dr. Bevilacqua's team received positive feedback from the U.S. Food and Drug Administration, as well as advice on their clinical development plan.
Amicrobe, Inc., currently has phase 1/2a clinical trials planned for Amicidin-α Surgical Gel and is working on several product variations with Amicidin-α, including Amicidin-α PFC (Prolonged Field Care) powder, which is being formulated specifically for use in military environments and is currently funded through the Medical Technology Enterprise Consortium by the U.S. Army Medical Research and Development Command. Amicidin-α gels, powders, and liquid solutions are being designed as cost-effective antimicrobial treatments and protective barriers for exposed tissue in surgical and traumatic wounds. If these products prove successful in clinical trials, they could offer new treatment measures to prevent wound infections due to surgery or combat trauma.
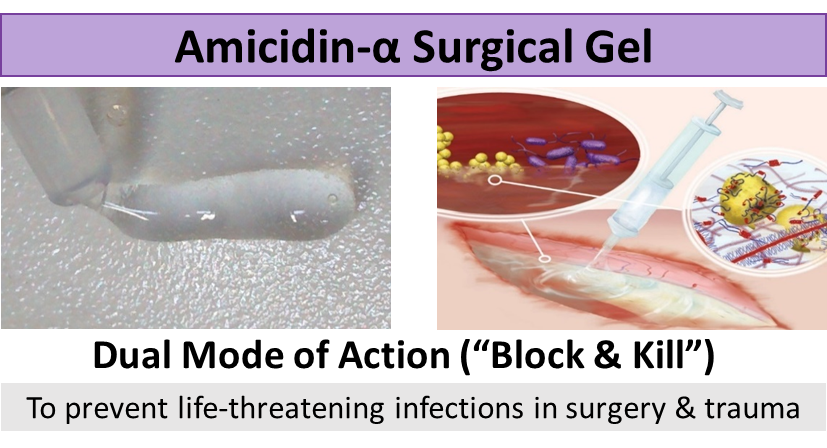
Figure 1: Amicidin-α Surgical Gel is formulated as a hydrogel to act as a barrier for wounds. The gel has antimicrobial properties to destroy and inhibit bacterial, viral, and fungal growth. This “block and kill” strategy allows Amicidin-α Surgical Gel to prevent infections in both traumatic wounds and surgical wounds.
Publications:
Bevilacqua, M., Huang, D., Wall, B., et al. 2017. Amino acid block copolymers with broad antimicrobial activity and barrier properties. Macromolecular Bioscience 17(10):1600492.
Links:
Last updated Thursday, November 13, 2025
 An official website of the United States government
An official website of the United States government
 ) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
