Peer Reviewed Cancer
Neutro-lizing Colorectal Cancer



Posted February 23, 2022
Triet Bui, Northwestern University

Mr. Triet Bui, Doctoral Student

Dr. Ronen Sumagin, Mentor
Colorectal cancer (CRC) remains the third most common cancer worldwide, with increasing incidence rates in younger populations. As such, guidelines recommend adults ages 45 and older for regular screening. At diagnosis, treatments can include surgical removal, chemotherapy, and much less commonly, radiation. Surgical resection occurs when the cancer has limited penetration into the wall of the large intestine and remains localized. If it metastasizes and the cancer reaches distant organ sites beyond the colon or rectum, chemotherapy such as FOLFOX (5-Fluorouracil, leucovorin, and oxaliplatin) is considered standard of care treatment.1 The relative 5-year survival rate for CRC is 64% following diagnosis; however, localized survival rate is 90%, while regional (spread to nearby lymph nodes) and distant stages drop to 71% and 14%, respectively.2
The tumor microenvironment consists of non-cancerous cells that envelop the growing cancer, which includes immune cells, structural cells (fibroblasts), and blood vessels. In cancer-related inflammation, immune cells that can infiltrate tumors such as the neutrophil, known as polymorphonuclear neutrophils (PMNs), have the ability to promote or destroy cancer cells. In work supported by a Fiscal Year 2019 Peer Reviewed Cancer Research Program Horizon Award, Triet Bui and colleagues expand upon this topic, delving into the mechanisms of PMNs and DNA damage repair in CRC using mouse models, human derived cell lines, and tissue samples.3
Previous findings contributed by Bui and others show PMNs infiltrate the innermost layer of the large intestinal wall, known as the mucosa, and cause DNA damage through double-strand breaks. This concurrently prevents DNA repair in the form of homologous recombination, resulting in genomic instability (increased tendency for DNA mutations).4 Current work by Bui and group show PMN suppression of homologous recombination in early-stage CRC tumor cells delays cancer growth, although in advanced stage CRC, PMNs promote an alternate form of DNA repair, non-homologous end joining. This leads to prolonged survival of the cancer cells. The important genes in this pathway are RAD51 for homologous recombination and Ku70 for non-homologous end joining.
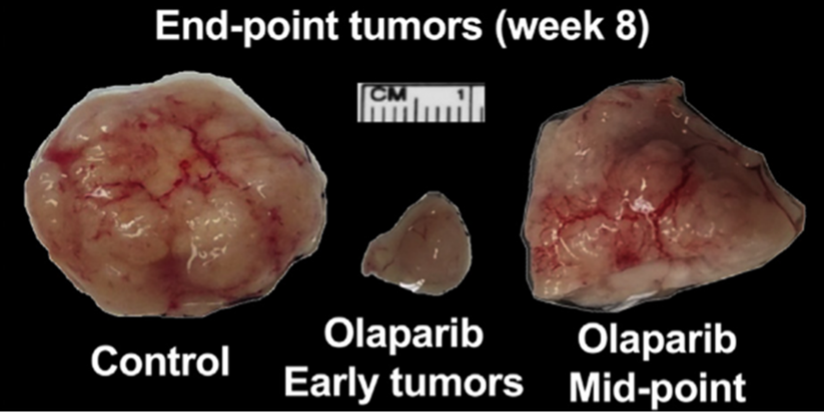
Figure 1: Representative images of suppressed mouse tumor development following the early and later timepoint Olaparib treatment regimen.
(Adapted from Bui, Gastroenterology. 2021).
The dichotomy of PMN function in early (suppressed homologous recombination and low non-homologous end joining) and advanced (suppressed homologous recombination and high non-homologous end joining) CRC presents a unique opportunity for new therapeutic intervention. Bui utilizes Olaparib, a Food and Drug Administration (FDA)-approved therapeutic to treat BRCA1/2 mutated breast cancers.5 BRCA1/2 are involved in the repair of DNA double-strand breaks through homologous recombination. Olaparib inhibits the enzyme that repairs DNA single-strand breaks and when used in BRCA 1/2 mutated cells, causes cancer cell death. Bui took advantage of the effectiveness against suppressed homologous recombination to show that Olaparib inhibited early CRC tumor development in mice. In contrast, Olaparib used later in CRC progression in mice had no effect on tumor growth or survival; however, non-homologous end joining inhibitors induced tumor lethality.
CRC regimens like chemotherapy only serve as a broad treatment option that targets rapidly dividing cancer cells. Unfortunately, this type of aggressive therapy can have limited success rates and toxicity to the patient. With the rising incidence of CRC among younger populations and the urgent need to develop novel therapeutics, this study bridges the gap by illustrating the benefit of repurposing FDA-approved drugs. Olaparib demonstrates efficacy in preclinical CRC models, which may translate into a faster therapeutic intervention for those suffering from early stage CRC. Further trials in human patients and determining other factors of PMN involvement in the tumor microenvironment are necessary, but Olaparib and the results of non-homologous end joining inhibitors may prove advantageous for the future of CRC.
References:
1 Clark JW. Patient education: Colorectal cancer treatment; metastatic cancer (Beyond the Basics). In: UpToDate, Post TW (Ed), UpToDate, Waltham, MA. (Accessed on November 08, 2021.)
2 American Cancer Society. Colorectal Cancer Facts & Figures 2020-2022. Atlanta: American Cancer Society; 2020.
3 Bui TM, Butin-Israeli V, Wiesolek HL, et al. 2021. Neutrophils alter DNA repair landscape to impact survival and shape distinct therapeutic phenotypes of colorectal cancer. Gastroenterology. 2021 Jul;161(1):225-238.e15. doi: 10.1053/j.gastro.2021.03.027. Epub 2021 Mar 19. PMID: 33753103.
4 Butin-Israeli V, Bui TM, Wiesolek HL, et al. 2019. Neutrophil-induced genomic instability impedes resolution of inflammation and wound healing. J Clin Invest. 2019 Feb 1;129(2):712-726. doi: 10.1172/JCI122085. Epub 2019 Jan 14. PMID: 30640176; PMCID: PMC6355304.
5 Robson M, Im SA, Senkus E, et al. 2017. Olaparib for metastatic breast cancer in patients with a germline BRCA mutation. N Engl J Med. 2017 Aug 10;377(6):523-533. doi: 10.1056/NEJMoa1706450. Epub 2017 Jun 4. Erratum in: N Engl J Med. 2017 Oct 26;377(17 ):1700. PMID: 28578601.
Links:
Last updated Monday, December 15, 2025
 An official website of the United States government
An official website of the United States government
 ) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
