Peer Reviewed Cancer



Posted September 3, 2021
Benedict Law, Ph.D., Joan & Sanford I. Weill Medical College of Cornell University
 Dr. Benedict Law
Dr. Benedict Law
Diffuse intrinsic pontine glioma (DIPG) also known as diffuse midline glioma is a fatal pediatric brain tumor. Due to the location of the tumor buried deep within the brain at the pons of the brain stem, the tumors are inoperable. The tumor location further complicates treatment because the blood brain barrier (BBB) prevents systemic therapeutics from entering the brain.1Radiotherapy offers short-term benefits in slowing down glioma progression; but is not curative.2Convection-enhanced delivery (CED), a minimally invasive method of placing catheters directly into the brain in order to bypass the BBB, has been used, but treatments are rapidly cleared from the brain before therapeutic effects can take place.3, 4New and effective drugs and drug delivery methods are required for the treatment of DIPG. Nanoparticles have been used to slow clearance time and distribution of drugs delivered via CED. Dr. Benedict Law at Weill Cornell Medicine developed a peptide nanofiber precursor (NFP) nanoparticle system that is comprised of individual peptide conjugates that self-assemble into a highly stable single-layer structure shown to improve tissue penetration.5, 6, 7In collaboration with Dr. Mark Souweidane, a pediatric neurosurgeon known for his translational and clinical research accomplishments for children with DIPG, Dr. Law’s team demonstrated that NFP nanoparticles, paired with CED, can improve access to the brain beyond the BBB.5With funding from a Fiscal Year 2016 Idea Award with Special Focus, Dr. Law and his collaborators evaluated the ability of NFPs to carry emtansine (DM1), a bio-conjugatable microtubule inhibitor approved as a drug for breast cancer treatment which has been shown to be effective against glioblastoma multiforme, via CED to treat orthotopically implanted human-derived DIPG tumors in mice.
Dr. Law and his team members, including Drs. Richard Ting and Vanessa Bellat, first investigated NFP nanoparticle retention properties within the pons of the brain stem. NFP nanoparticles were conjugated to a radioisotope (Zr-NFP) and imaged using positron emission tomography and computed tomography (PET/CT), which showed NFP nanoparticles had prolonged retention times in the brain. The Zr-NFP nanoparticles alone were non-toxic to normal brain cells. Transmission electron microscopy confirmed that Zr-NFP successfully infused into the pons, and PET/CT imaging analyses revealed that Zr-NFP remained at the site of infusion in the pons for more than 3 weeks post-CED. The half-life of the NFP within the pons was 60.3 days, while the positive control compound was cleared within 4 hours. Of the original infused dose of Zr-NFP, 47% was found in situ after 4 days, and 38% remained after 21 days, with minimal amounts accumulating in other organs. Comparatively, only 4% of the original dose of the positive control compound remained in the brain 4 days after infusion. After observing the benefits of using NFP nanoparticles, the investigators went on to evaluate the use of CED in delivering DIPG chemotherapeutic agents conjugated with NFP.
Fluorescence microscopy and imaging studies demonstrated that multiple glioma cell lines uptake DM1-loaded NFP (DM1-NFP) delivered via CED and that the uptake of DM1-NFP increased proportionally with time. Cell viability assays demonstrated that DM1-NFP was cytotoxic to all glioma cell lines tested and 10-30 times less potent against normal brain cells like astrocytes, suggesting selective toxicity toward cancer cells. Taken together, the retention of NFP nanoparticles in the pons and the potency of DM1-NFP against DIPG cell lines demonstrate the feasibility of using NFP as a nanocarrier of DM1 for in vivo studies.
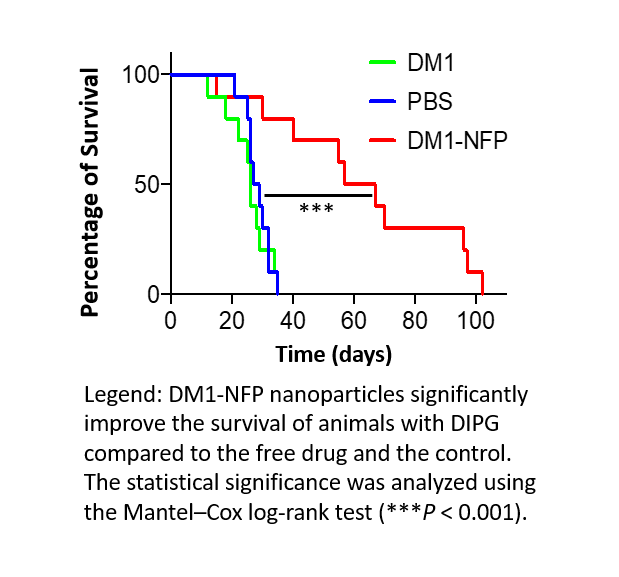
To test DM1-NFP in vivo, analysis of the therapeutic efficacy of DM1-NFP delivered via CED was conducted by implanting two different DIPG cell lines into the pons of non-obese diabetic/severe combined immunodeficiency gamma (NSG) mice. Analysis of the percentage of the injected dose (ID) of DM1 7 days post-treatment showed that 17% of the ID was present in animals treated with DM1-NFP compared to only 2% in animals treated with DM1 alone. Gliomas in mice treated with DM1-NFP delivered via CED grew significantly slower than tumors in mice treated with DM1 or PBS alone. Importantly, the median survival of animals treated with DM1-NFP was 62 days, compared to 26 and 28 days in mice treated with unconjugated DM1 and PBS, respectively. Histology confirmed that gliomas were significantly smaller in animals treated with DM1-NFP compared to animals treated with only DM1 and PBS. These results demonstrate that when DM1 is conjugated with NFP and delivered by CED there is prolonged local retention of DM1-NFP in the brain and enhanced anti-glioma activity compared to unconjugated DM1 alone.
Dr. Law and his colleagues showed that use of NFP nanocarriers minimized off-target delivery and prolonged retention of DM1 at the site of CED infusion. DM1-NFP exhibited selective toxicity toward glioma cells, and a single treatment increased survival times in mice. The results of this study are a critical first step in identifying effective treatment methods for DIPG. The NFP system offers numerous advantages, including its efficacy with low drug concentrations, clinical translatability, and its reduction of toxicity of systemically delivered drugs. Importantly, Dr. Law’s NFP approach may help to resolve issues with drugs that were previously abandoned for treatment of DIPG, such as poor solubility and delivery or toxicity, opening up new avenues of chemotherapeutic options for patients. Future studies investigating other drugs, dosing levels, and ideal CED parameters will aid in the optimization of treatment and identify other potential therapeutics for this difficult to treat disease.
References:
1 Zhou Z, Singh R, and Monje M. 2017. Diffuse intrinsic pontine glioma: New pathophysiological insights and emerging therapeutic.Current Neuropharmacology 15(1):88-97.
2 Vanan MI and Eisenstat DD. 2015. DIPG in children – What can we learn from the past?Frontiers in Oncology&5:237.
3 Sewing AC, Caretti V, Lagerweij T, et al. 2014. Convection enhanced delivery of carmustine to murine brainstem: A feasibility study.Journal of Neuroscience Methods 238:88-94.
4 Singleton WGB, Bienemann AS, Woolley M, et al. 2018. The distribution, clearance, and brainstem toxicity of panobinostat administered by convection-enhanced delivery.Journal of Neurosurgery: Pediatrics 22(3):288-296.
5 Bellat V, Ting R, Southard, TL, et al. 2018. Functional peptide nanofibers with unique tumor targeting and enzyme-induced local retention properties.Advanced Functional Materials 28(44):1803969.
6 Tosi U, Kommidi, H, Bellat V, et al. 2019. Real-time, in vivo correlation of molecular structure with drug distribution in the brain striatum following convection enhanced delivery.ACS Chemical Neuroscience 10(5):2287-2298.
7 Cariou PL, Lee KL, Pokorski, JK, et al. 2016. Diffusion and uptake of tobacco mosaic virus as therapeutic carrier in tumor tissue: Effect of nanoparticle aspect ratio.The Journal of Physical Chemistry B 120(26):6120-6129.
Link:
Last updated Wednesday, September 17, 2025
 An official website of the United States government
An official website of the United States government
 ) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
