Peer Reviewed Cancer



Posted October 2, 2019
Catherine Bollard, M.D., Children's Research Institute at Children's National Medical Center (CNMC)
Award Mechanism: Idea Award with Special Focus

Dr. Catherine Bollard
Scientists have found that several types of cancers exhibit similar genetic signatures where the transforming growth factor-beta (TGF-ß) pathway modulates the activation and function of natural killer (NK) cells.1 The TGF-ß pathway can modulate immune suppression by regulating NK cells that target and remove virus-infected and malignant cells and affect the recruitment of other immune cells.1,2,3 However, cancer cells can become resistant to TGF-ß’s regulation, while capitalizing on the pathway’s secondary functions of stimulating cellular transformation and encouraging the formation of new blood vessels, which promote tumor growth.1,4 While donor hematopoietic cell transplantation of peripheral blood NK cells has been used to treat acute myelogenous leukemia with positive clinical outcomes, using donor cells poses a risk of developing an autoimmune response, such as graft-versus-host disease (GVHD), that is caused by the innate immune system attacking the donor cells.5 Using a patient’s own NK cells is ideal, but cancer patients’ peripheral blood NK cells are generally scarce, with limited killing activity in the tumor environment, and have a short longevity of approximately 2 weeks.5
Dr. Catherine Bollard was awarded a FY14 Idea Award with Special Focus with Drs. Conrad Russell Cruz and Eric Yvon. They theorized that it was possible to boost the immune system by using NK cells extracted from frozen donor umbilical cord blood and to engineer the extracted NK cells to be resistant to TGF-ß modulated inhibition. They proposed that NK cells from cord blood can be more easily isolated and expanded, are persistent after infusion into patients, and pose less risk of provoking GVHD. The team focused on addressing neuroblastoma first and noted the versatility of the strategy for other cancers with this NK cell inhibiting TGF-ß signature if successful.
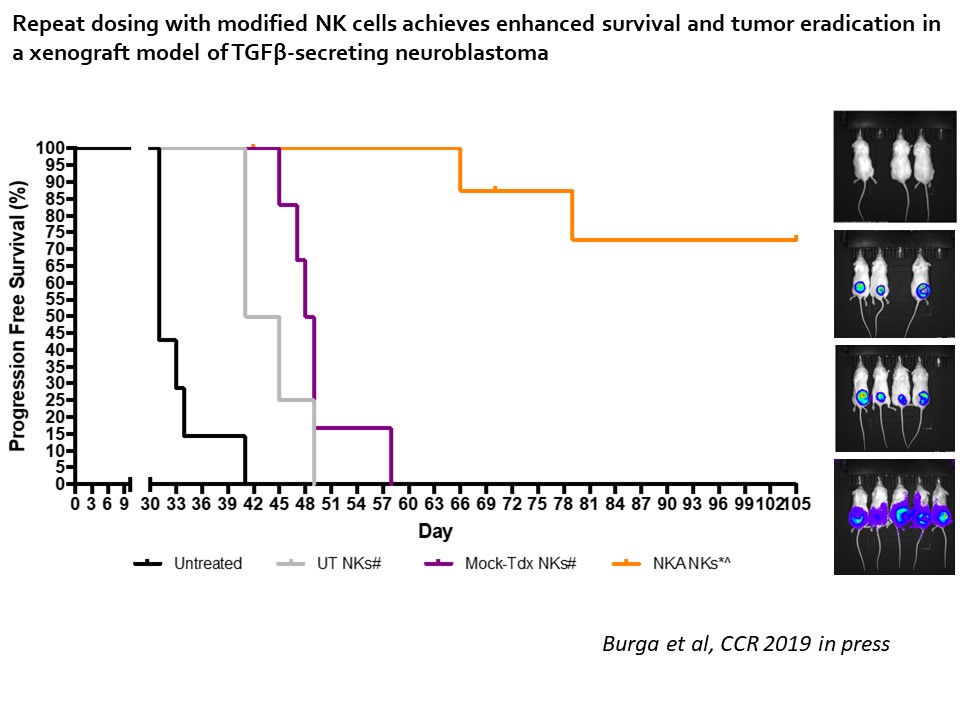
First, Dr. Bollard and colleagues extracted and expanded NK cells from cord blood from donor banks and examined the cells’ function and phenotype. They reported that expanded cord NK cells had similar cytotoxicity effects on human myelogenous leukemia cells in vitro as fresh NK cells extracted from healthy, peripheral blood donors, confirming the practicality of using cord NK cells. Dr. Bollard’s team then transduced cord NK cells, engineering them to express a mutant TGF-ß receptor, protecting the cells from TGF-ß-related inhibitory effects. Her team confirmed the NK cell modification molecularly through western blot and flow cytometry, and they reported greater success with engineering cord NK cells than with peripheral blood NK cells. Her team then examined the functionality of the cells by incubating either human myelogenous leukemia cells or neuroblastoma cells with the modified cord NK cells in vitro. The group noted increased cytotoxic activity and cytotoxic effect from the modified cord NK cells compared to unmodified cord NK cells, establishing confidence in the effectivity of the engineered cord NK cells. Lastly, to test translatability of engineered NK cells’ effect in vivo, Dr. Bollard’s team developed a mouse xenograft model to grow tumors of human neuroblastoma cells. The group reported success in reducing tumor growth in mouse xenograft models that were treated with modified cord NK cells for the duration of the study (105 days), extending mice longevity to approximately two times longer than mice models treated with unmodified cord NK cells and more than three times longer than untreated mice models. This work was published in Clinical Cancer Research in 2019. 6
Overall, this PRCRP funded project was able to yield positive results that will now enable Dr. Bollard’s team to move toward the next step. Dr. Bollard plans to seek regulatory approval to grow larger quantities of TGF-ß-resistant cord blood derived NK cells for a pilot clinical trial to treat patients with advanced neuroblastoma after autologous stem cell transplantation. If successful in clinical trials, this method of engineering NK cells extracted from umbilical cord blood to eliminate cancer cells could potentially address multiple types of cancers that exhibit high TGF-ß and low NK cell function including, but not limited to, myeloma, melanoma, mesothelioma, pancreatic cancer, and hematologic cancers. The impact of a broad spectrum immunotherapy would be a paradigm shift in cancer treatment, leading to the development of a new first line of defense against cancer compared to more expensive, personalized therapies that target specific types of malignancies.
References:
- Haque S and Morris JC. 2017. Transforming growth factor-β: A therapeutic target for cancer. Hum Vaccin Immunother. Aug 3;13(8):1741-1750.
- Fessenden TB, Duong E, and Spranger S. 2018. A team effort: Natural killer cells on the first leg of the tumor immunity relay race. J Immunother Cancer. Jul 9;6(1):67.
- Liu M, Zhou J, Chen Z, and Cheng AS. 2017. Understanding the epigenetic regulation of tumours and their microenvironments: Opportunities and problems for epigenetic therapy. J Pathol. Jan;241(1):10-24.
- Fabregat I, Fernando J, Mainez J, and Sancho P. 2014. TGF-beta signaling in cancer treatment. Curr Pharm Des. 20(17):2934-2947.
- Lowry LE and Zehring WA. 2017. Potentiation of natural killer cells for cancer immunotherapy: A review of literature. Front Immunol. Sep 1;8:1061.
- Burga RA, Yvon E, Chorvinsky E, Fernandes R, Cruz CR, Bollard CM. 2019. Engineering the TGFβ receptor to enhance the therapeutic potential of natural killer cells as an immunotherapy for neuroblastoma. Clin Cancer Res.
Last updated Wednesday, March 19, 2025














