


Posted September 29, 2016
Edward H. Hinchcliffe, Ph.D., University of Minnesota, Twin Cities

Edward H. Hinchcliffe, Ph.D., University of Minnesota, Twin Cities
Pediatric brain tumors, including high-grade gliomas like diffuse intrinsic pontine glioma and glioblastoma multiform, are the leading cause of cancer-related death in people under the age of 20 and the third leading cause of cancer-related death in those ages between 20-39 years old. Sequencing studies of multiple tumor samples have identified a set of proteins that are commonly mutated in high-grade pediatric brain tumors: H3.3, ATRX, DAXX, and p53. The first three proteins form a complex with each other and are responsible for remodeling DNA when tightly packed into the configuration known as chromatin, which further compacts to form chromosomes. The fourth, p53, is responsible for regulating many cellular functions, but one of the most studied is its role in initiating programmed cell death in response to DNA damage.
Leading up to receiving his Fiscal Year 2013 (FY13) Idea Award with Special Focus, Dr. Edward Hinchcliffe made preliminary observations that the trio of H3.3/ATRX/DAXX may also play a key role to ensure that DNA separates appropriately during cellular division. So far, under this award, Dr. Hinchcliffe has made great progress in understanding the mechanisms that drive this process and how defects in DNA segregation during cell division may lead to tumorigenesis.
A normal, non-reproductive human cell contains 23 pairs of chromosomes. As a cell prepares to divide via a process called mitosis, a duplicate of each chromosome is made. The mechanisms guiding cellular division are highly regulated to ensure that each new daughter cell contains one complete set of chromosomes. When the chromosomes missegregate, both cells end up having an incorrect number of chromosomes and become what is called aneuploid. One of the many checkpoints cells use to ensure that chromosomes are separating appropriately consists of modifications to the amino acid serine number 31 (S31) on one of the proteins in the trio, H3.3, which has been the focus of Dr. Hinchcliffe's studies to date. Dr. Hinchcliffe and his team found that if chromosome(s) starts to misalign during mitosis, H3.3 S31 becomes highly modified on the misaligned chromosome. In this way, S31 modification appears to act as a Proximity Sensor for appropriate chromosome alignment. After mitosis is complete, the S31 modification is amplified and becomes evident on other chromosomes, not just the misaligned one(s).
Dr. Hinchcliffe poses the novel hypothesis that these S31 modifications are sensed by ARTX and DAXX. Little is known about these putative mechanisms, which is an active area of exploration for Dr. Hinchcliffe. However, he did find that the H3.3 S31 modifications result in activation of p53. In cells that were identified to have chromosomes being missegregated during mitosis, he injected antibodies into those specific cells at precise time points to block the cell's ability to modify S31. Using these advanced techniques, Dr. Hinchcliffe was able to confirm that p53 was directly activated due to the H3.3 S31 marker of missegregated chromosomes. These are the first studies describing such a Proximity Sensor for missegregated chromosomes, and they provide novel insight into how this sensor may function to prevent any further cell growth and replication.
In summary, these studies shed some important light onto how defects in H3.3, the Proximity Sensor, can drive tumorigenesis and cancer progression. The observed mutations in H3.3 in glioblastoma patient tumors could either result in incorrect S31 modifications or change the interfaces that are required for ARTX and DAXX to interact with H3.3. Either mechanism results in the cell's inability to propagate the p53 activation signal, allowing the defective cell to proliferate. This study provides the knowledge foundation crucial for designing more advanced diagnostic tools to detect brain tumors at earlier stages and to develop new, effective treatments for the identified tumors.
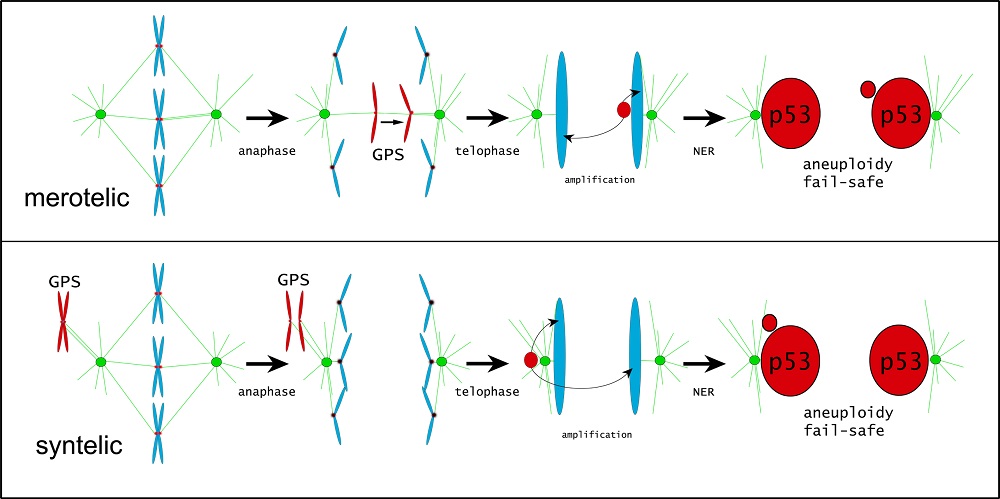
Mechanisms by which chromosomes can missegregate and proposed model of a Proximity Sensor for appropriate chromosome alignment
Publication
Hinchcliffe EH, Day CA, Karanjeet KB, et al. 2016. Chromosome missegregation during anaphase triggers p53 cell cycle arrest through histone H3.3 Ser31 phosphorylation. Nat Cell Biol 18(6):668-675.Link:














