Prostate Cancer
Neuroendocrine Differentiation and Enzalutamide Resistance in Prostate Cancer



Posted July 8, 2022
Isaac Yi Kim, M.D., Ph.D., MBA, Rutgers, State University of New Jersey

Dr. Isaac Yi Kim
In men with castration-resistant prostate cancer (CRPC), second-line antiandrogen therapy (SAT) has been used as a cornerstone in the standard of care. Currently, there are four US FDA-approved SAT agents: enzalutamide, apalutamide, darolutamide, and abiraterone. Nevertheless, clinical benefits are limited because not all prostate cancers are vulnerable to these therapies. In some cases, more aggressive types of prostate cancer, such as neuroendocrine prostate cancer, emerge and do not respond to SAT. For other patients, their tumors initially respond to SAT, but the tumors become resistant to treatment. Previous studies had led researchers to determine that the process of neuroendocrine differentiation, where prostate cancer cells characteristically change into neuroendocrine-like cells, occurred independently from androgen signaling. But recent preliminary data obtained by Dr. Isaac Kim's team at Rutgers, State University of New Jersey, suggests for the first time that these two pathways may be interconnected. Specifically, they found that the TCF4 gene, which codes for a transcription factor, may be important in mediating neuroendocrine differentiation in response to enzalutamide treatment. With the support of a FY16 Idea Development Award, the team sought to further elucidate the mechanism by which TCF4 regulates neuroendocrine differentiation and induces enzalutamide resistance in CRPC.
Through this work, Dr. Kim and his team found that the neuroendocrine markers, such as chromogranin A and parathyroid hormone related peptide (PTHrP), are highly expressed in enzalutamide-resistant human prostate cancer cells. After further analysis of these neuroendocrine differentiation related genes, they determined that enzalutamide treatment causes TCF4 to interact with a gene called beta-catenin, and that this interaction mediates neuroendocrine differentiation. The role of TCF4 in stimulating these neuroendocrine differentiation pathways was confirmed in tissues obtained from men who died of metastatic prostate cancer, which demonstrated a positive correlation between the expression levels of TCF4 and the PTHrP neuroendocrine marker. Next, Dr. Kim's team studied the relationship between TCF4 and enzalutamide resistance in mouse xenograft models. Their results demonstrated a reciprocal connection between enzalutamide resistance and neuroendocrine differentiation. When TCF4 was overexpressed, it caused enzalutamide resistance in the mouse model. Conversely treatment of the mice with enzalutamide increased TCF4 expression levels by stimulating expression of the androgen receptor splice variant AR-V7, which is known to be associated with enzalutamide resistance. Collectively, these results demonstrate a clear relationship between enzalutamide treatment and neuroendocrine differentiation.
With this new potential therapeutic target identified, Dr. Kim's team wanted to try targeting the TCF4/PTHrP axis. The team tested a TCF4 inhibitor, PKF-118-310, and/or a PTHrP antagonist in an enzalutamide-resistant mouse model and found that both compounds decreased tumor growth over a seven week period. However, there was no synergistic effect between PKF118-310 and the PTHrP antagonist, meaning that these treatments do not provide an increased effect when combined compared to when they are given individually. These results collectively suggest that TCF4 and PTHrP inhibitors may be an effective treatment option for enzalutamide-resistant prostate cancer, and Dr. Kim's team is currently working to develop additional small molecules that target TCF4 to provide new treatments for men with CRPC.
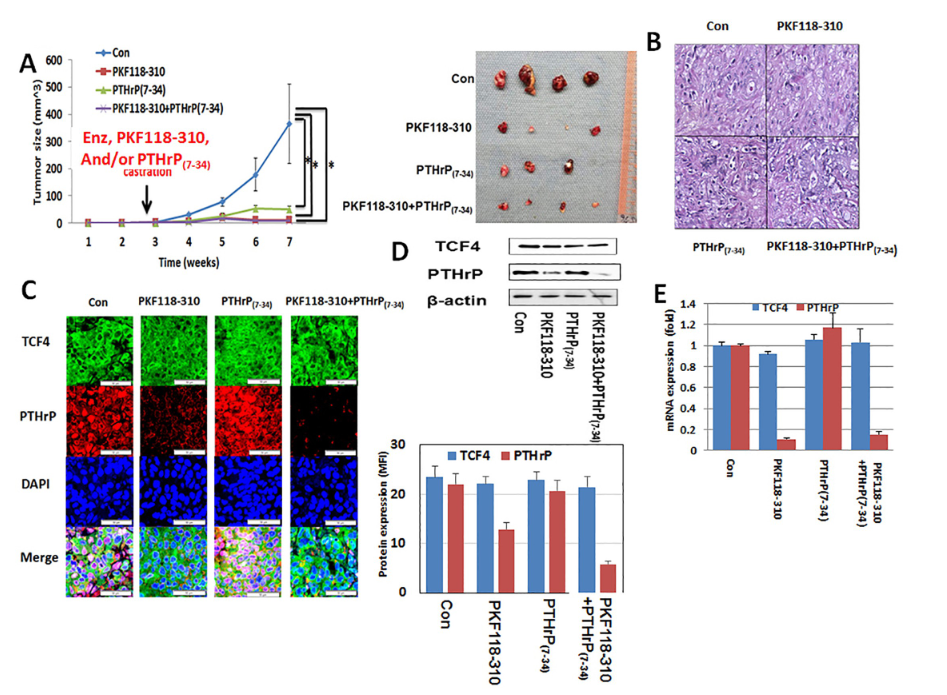
Figure 1: After injection of LNCaP-EnzR into the flanks of forty Rag2−/−, γc−/− immunodeficient mice, all mice were surgically castrated divided into four groups of ten each. Animals in predesignated groups were treated daily with PKF118-310 (0.85 mg/kg intraperitoneal) and/or PTHrP(7- 34) (0.2 mg/kg subcutaneous). All mice were administered daily 10 mg/kg enzalutamide orally. A. Treatment of PKF118-310 and/or PTHrP(7- 34) with 10 mg/kg enzalutamide decreased tumor growth compare with vehicle treatment control group (con). B. H&E staining. There was no difference among all groups. C. Immunofluorescence staining for TCF4 (green), PTHrP (red) with DAPI (blue) staining. Consistent with its mechanism of action, PFK118-310 treatment decreased PTHrP protein levels. However, there was no effect on TCF4 levels. In contrast, PTHrP(7-34) had no demonstrable effect on the protein levels of both TCF4 and PTHrP. Treatment of PKF118-310 decreased PTHrP protein (D) and mRNA expression (E). Error bars indicate average - SE and * p-value<0.05.
Publication:
Lee GT, Rosenfeld JA, Kim WT, Kwon YS, Palapattu G, Mehra R, Kim WJ, Kim IY. TCF4 induces enzalutamide resistance via neuroendocrine differentiation in prostate cancer. PLoS One. 2019 Sep 19;14(9):e0213488. doi: 10.1371/journal.pone.0213488. PMID: 31536510; PMCID: PMC6752758.
Link:
Neuroendocrine Differentiation and Enzalutamide Resistance in Prostate Cancer
Last updated Wednesday, September 17, 2025
 An official website of the United States government
An official website of the United States government
 ) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
