Prostate Cancer
Chronic inflammation promotes prostate cancer initiation, progression, and recurrence



Posted March 29, 2017
Angelo De Marzo, M.D., Ph.D.; Johns Hopkins University, Elizabeth Platz, M.P.H., Sc.D.; Johns Hopkins University, Karen Sfanos, Ph.D.; Johns Hopkins University, Andrew Goldstein, Ph.D.; University of California, Los Angeles

Photo provided by Johns Hopkins University

Photo provided by University of California, Los Angeles
An inflammatory microenvironment is a critical component driving tumorigenesis, and a number of cancers, potentially including prostate cancer (PCa), have been shown to initiate in sites of chronic inflammation. Dr. Angelo De Marzo and colleagues at Johns Hopkins University , including William G. Nelson M.D., Ph.D., in a number of early groundbreaking studies suggested that proliferative signals from inflammatory cells could help lead to PCa initiation. These studies, supported in part from a PCRP FY05 Idea Development Award, helped to establish chronic inflammation as a risk factor for PCa. Although this seminal work showed a clear connection between chronic inflammation and PCa, exact mechanisms of how inflammation contributes to PCa tumorigenesis are still unknown. Further understanding of this process could lead to the discoveries of important biomarkers for PCa progression or to novel prevention or intervention strategies.
In joint efforts between the laboratories of Drs. Angelo De Marzo, Elizabeth Platz, and Karen Sfanos, this interdisciplinary team at Johns Hopkins University sought to examine the connection between a specific innate immune cell type called mast cells and risk of PCa recurrence. With PCRP funding from a FY11 Population-Based Research Award (Platz) and a FY13 Health Disparity Research Award (Sfanos), they analyzed the number of mast cells in benign and tumor tissues from the PCRP-funded Prostate Cancer Biorepository Network and the associated risk of cancer recurrence. The study was spearheaded by Heidi Hempel, B.S., a Nested Trainee on Sfanos' PCRP award and a Pathobiology graduate student at the Johns Hopkins School of Medicine. Interestingly, the team found that risk of recurrence was associated with low mast cell density in the tumor tissue yet high mast cell density in the normal tissue. These results suggest that intratumoral mast cells may protect against PCa and could serve as a potential biomarker to predict recurrence.
Furthermore, Dr. Sfanos and her team expanded these studies to look at the mast cell numbers in tissues of African American men who have greater PCa incidence and mortality. Through this work, they hope to use these tools to understand if differential presence or abundance of mast cells can predict disease aggressiveness or explain why PCa is often more aggressive in African American men. Understanding the consequences of intraprostatic inflammation or its causes could serve as a viable primary prevention strategy for this high-risk population.
Building off of the work done by the Hopkins group, Andrew Goldstein and his team at UCLA began looking at the potential involvement of progenitor cells in inflammation and PCa initiation. Prostate epithelium consists of distinct layers made up of three cell types: basal, luminal, and neuroendocrine cells. Previous studies in human prostate tissue had found that progenitor cells in the basal layer are common but progenitor cells in the luminal layer are rare. With support from a PCRP FY12 Idea Development Award, Dr. Goldstein and his laboratory developed a strategy to track down previously undetectable progenitor cells in the luminal layer based on their low expression of a molecule called CD38. Utilizing tissue microarray samples, they examined luminal progenitor cells with low expression of CD38 and found them clustered around areas of inflammation. They determined that these progenitor cells have the capacity to divide and expand; giving them the ability to respond to genetic changes and develop into PCa. Therefore, these studies suggest that chronic inflammation likely increases risk for PCa because it increases the number of progenitor cells that can initiate cancer.
Additional work is ongoing to isolate and profile these progenitor cells from areas of inflammation in order to understand what makes them capable of expanding and responding to genetic changes. Dr. Goldstein and his group believe that the low expression of this molecule may provide an advantage to these progenitor cells that is maintained in the most aggressive prostate tumors that also exhibit low expression of CD38. These collective results support inflammation-associated luminal cells as a target cell for aggressive PCa.
Taken together, these results indicate that chronic inflammation is likely to play an important role in all stages of PCa growth from tumor initiation to disease recurrence after therapy. Furthermore, these findings give us a deeper understanding of the inflammatory mechanisms that contribute to PCa development and aggressiveness that were previously unknown. Although more research needs to be done in this area, this collective work supported by the Prostate Cancer Research Program serves as an important step forward to translating these findings into the clinic in order to develop new prevention and treatment strategies for PCa.
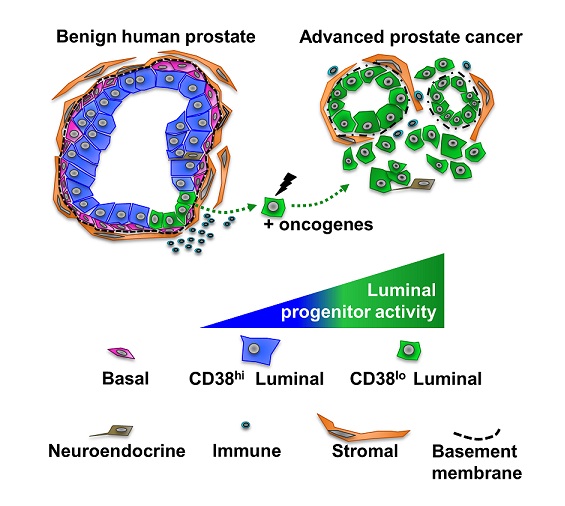 Goldstein Figure Legend: CD38-lo luminal cells (green) isolated from benign human prostate tissue have significantly greater progenitor activity than isolatedCD38-hi luminal cells (blue). CD38-lo luminal cells are expanded in glands adjacent to inflammation and can be transformed by oncogenes to initiate aggressive human prostate cancer. Advanced prostate tumors with an increased likelihood of biochemical recurrence and metastasis exhibit a CD38-lo luminal phenotype. From Liu et al, Cell Reports (2016).
Goldstein Figure Legend: CD38-lo luminal cells (green) isolated from benign human prostate tissue have significantly greater progenitor activity than isolatedCD38-hi luminal cells (blue). CD38-lo luminal cells are expanded in glands adjacent to inflammation and can be transformed by oncogenes to initiate aggressive human prostate cancer. Advanced prostate tumors with an increased likelihood of biochemical recurrence and metastasis exhibit a CD38-lo luminal phenotype. From Liu et al, Cell Reports (2016).
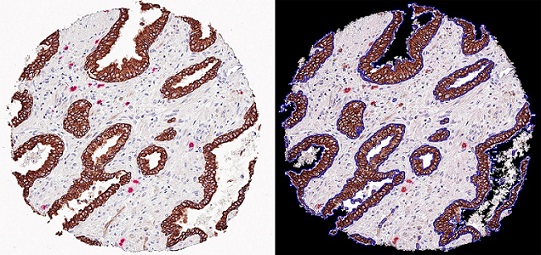
Sfanos Mast cell TMA spot: The image on the left is a tissue microarray (TMA) spot stained for cytokeratin-8 (brown) and mast cell trypase (red). On the right is a TMA spot analyzed with image analysis software that calculates epithelial area and mast cell counts.
Publications:
Public and Technical Abstract:
Interactions between Dietary Factors and Inflammation in Prostate Carcinogenesis
Infections and Innate Immunity in Prostate Cancer Racial Disparities
Last updated Monday, March 10, 2025














