Prostate Cancer
Power Outage: Targeting Prostate Cancer Metabolism



Posted July 1, 2016
Daniel Frigo, Ph.D., University of Houston
 Prostate cancer (PCa) is the second leading cause of cancer-related deaths in men. The androgen receptor (AR) signaling cascade is known to be involved in the progression to the deadly advanced stages of the disease. Many current therapies for PCa attempt to block the AR signaling pathway, although the tumors eventually become resistant to these therapies as the AR finds alternative mechanisms of activation; thus new therapies are greatly needed. Further understanding of the signaling pathways downstream of the receptor can help identify novel drug targets to develop new, improved treatments for PCa.
Prostate cancer (PCa) is the second leading cause of cancer-related deaths in men. The androgen receptor (AR) signaling cascade is known to be involved in the progression to the deadly advanced stages of the disease. Many current therapies for PCa attempt to block the AR signaling pathway, although the tumors eventually become resistant to these therapies as the AR finds alternative mechanisms of activation; thus new therapies are greatly needed. Further understanding of the signaling pathways downstream of the receptor can help identify novel drug targets to develop new, improved treatments for PCa.
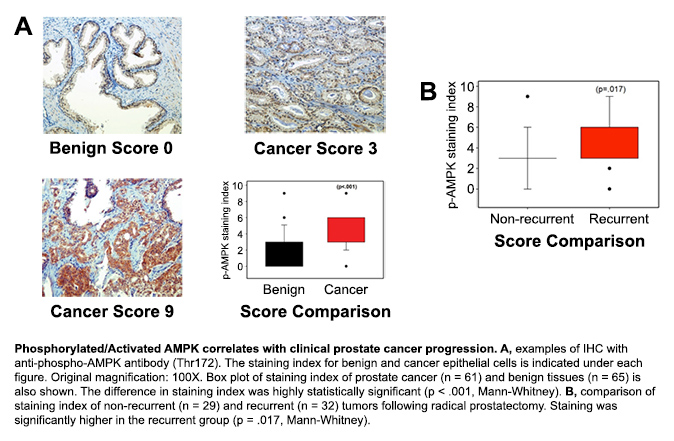
With support from a FY11 PCRP Idea Development Award, Dr. Daniel Frigo and his research team at the University of Houston demonstrated a key role for two metabolic proteins that fuel PCa cell growth through the AR pathway: CAMKKbeta and AMPK. Using a combination of radiolabeled assays and emerging metabolic approaches, they discovered that PCa cells respond to androgen treatment by increasing rates of glucose and fatty acid oxidation as well as overall glucose metabolism, and that these changes were dependent on androgen-mediated CaMKKbeta and AMPK activity. Their results further indicated that the AMPK-mediated metabolic changes occurred through several downstream cellular processes, affording distinct growth advantages to the prostate cancer cells. Importantly, CaMKKbeta was shown to be required for advanced, castrate-resistant PCa growth. Several other laboratories have also confirmed CaMKKbeta as a biomarker for both early and advanced stage PCa, which complements Dr. Frigo's findings and further suggests activated AMPK levels correlate with disease progression.
Taken together, the findings from Dr. Frigo's lab demonstrate that androgens co-opt the CaMKKbeta-AMPK signaling cascade to increase PCa cell growth. The current study points to the potential utility of developing metabolic-targeted drug therapies to shut down the power source for PCa cell growth and supports efforts to continue translating these findings towards the clinic. Dr. Frigo and his team recently received a 5-year R01 grant from the NIH, which will allow them to expand upon the results from their PCRP-funded study by identifying additional downstream targets of CaMKKbeta-AMPK signaling and other oncogenic pathways that appear to coordinate with CaMKKbeta-AMPK signaling to drive cancer progression.
Public and Technical Abstracts:
Research Links:














