Prostate Cancer
Deciphering the Adipocyte-Tumor Cell Interaction in Prostate Cancer



Posted October 11, 2016
Maria Diaz-Meco, Ph.D., and Jorge Moscat, Ph.D., Sanford-Burnham Medical Research Institute, La Jolla, CA

Maria Diaz-Meco, Ph.D., and Jorge Moscat, Ph.D., Sanford-Burnham Medical Research Institute, La Jolla, CA
Prostate cancer (PCa) is one of the leading causes of death among men in the United States. Obesity is another growing epidemic and represents one of the greatest threats to global human health. Epidemiological studies have shown a clear correlation between obesity and poor prognosis in many cancers, including PCa. However, little is known about the molecular basis underlying this connection and biochemical and genetic studies using physiologically relevant models that mimic the complexity of these processes are sorely needed.
Through collaborative efforts, Drs. Maria Diaz-Meco and Jorge Moscat from Sanford-Burnham Medical Research Institute, sought to deduce the mechanisms connecting obesity and PCa and identified the protein p62/SQSTM1 as a critical regulator of obesity. With support from a FY12 Synergistic Idea Development Award, they expanded this discovery by developing a PCa mouse model with p62 adipocyte deficiency to examine the interaction between obesity and PCa. Surprisingly, p62 deficiency did not cause obesity in this mouse model, suggesting the tumor was impacting the metabolism of the mice. Through further metabolic characterization, they revealed that these mice were undergoing cancer-associated cachexia, a condition displaying progressive weight loss in response to malignant growth. The loss of p62 also had a significant impact on PCa tumorigenesis. Drs. Diaz-Meco and Moscat discovered that p62 deficiency in adipose tissue led to higher incidences of aggressive types of PCa compared to control mice. Furthermore, these mice displayed higher incidences of metastases and interestingly, a difference in tissue preference for metastasis with increased tumors arising in the liver and lung. These results implicate that p62 loss in adipocytes may be a driver of PCa tumorigenesis and metastasis.
Taken together, this work reveals important roles of p62 in the interaction between adipocytes and tumor cells and demonstrates a direct molecular link between obesity and PCa progression in mice. In future studies, Drs. Diaz-Meco and Moscat plan to further decipher the mechanism by which the adipose tissue controls PCa and how interactions between the tumor and adipose tissue lead to cachexia. Through this work, they hope to show that manipulating p62-derived signals can be used to treat prostate tumors by preventing the metabolic impact of obesity.
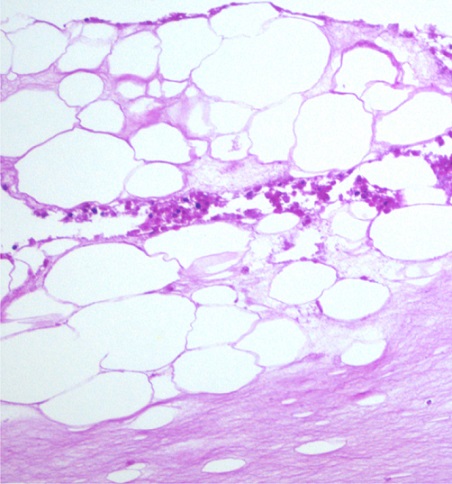
Adipocytes promote the invasion of prostate cancer cells
Public and Technical Abstracts:
Targeting the Adipocyte-Tumor Cell Interaction in Prostate Cancer Treatment
Research Links:
Last updated Thursday, December 5, 2024














