Prostate Cancer
Development of Novel Drugs That Target Coactivation Sites of the Androgen Receptor for Treatment of Antiandrogen-Resistant Prostate Cancer



Posted April 19, 2016
Dr. Artem Cherkasov, University of British Columbia
PC111132
 Prostate cancer (PCa) is the most frequently diagnosed non-cutaneous male cancer and the second leading cause of male cancer death. If diagnosed early, it is usually curable by surgery and/or radiotherapy. However, after primary treatment, prostate cancer recurs in 20-40% of patients with high-grade disease. The primary driver and drug target in recurrent prostate cancer is the Androgen Receptor (AR), which can be directly inhibited with drugs such as Bicalutamide and Enzalutamide. Because many tumors adapt and develop resistance to these drugs, there is still an urgent need to create new types of AR inhibitors that target this protein by entirely different mechanisms and to which PCa does not readily develop resistance.
Prostate cancer (PCa) is the most frequently diagnosed non-cutaneous male cancer and the second leading cause of male cancer death. If diagnosed early, it is usually curable by surgery and/or radiotherapy. However, after primary treatment, prostate cancer recurs in 20-40% of patients with high-grade disease. The primary driver and drug target in recurrent prostate cancer is the Androgen Receptor (AR), which can be directly inhibited with drugs such as Bicalutamide and Enzalutamide. Because many tumors adapt and develop resistance to these drugs, there is still an urgent need to create new types of AR inhibitors that target this protein by entirely different mechanisms and to which PCa does not readily develop resistance.
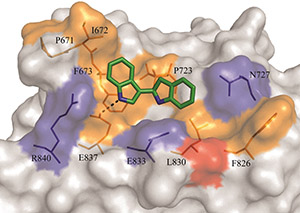
To tackle this problem, Dr. Artem Cherkasov and his team at the University of British Columbia are identifying novel drug candidates that target previously unrecognized regulatory sites of the AR. With support from a FY11 Idea Development Award, they identified a new regulatory site on the AR called BF3, which had not been fully characterized, although there was some evidence for its involvement in AR activation. Additionally, they performed rigorous computer-aided drug design and developed specific functional screening assays to demonstrate that direct binding of select compounds to the BF3 site could lead to effective inhibition of AR transcriptional activity. The potent novel AR inhibitors developed by Dr. Cherkasov's team also demonstrated effectiveness against drug-resistant forms of PCa. These ground-breaking results validate the BF3 site as an excellent AR drug target and demonstrate an intriguing possibility of direct inhibition of the co-factor/co-regulator recruitment to the AR, as opposed to traditional indirect inhibition through competition with endogenous androgens for the AR ligand-binding site.
In contrast to large scale drug screening done by pharmaceutical companies, which typically yield less than a 0.03% success rate, Dr. Cherkanov's computer-aided approach achieves a success rate as high as 30-50%. Dr. Cherkanov plans to continue advancing the development of these unique BF3-directed AR inhibitors with further pharmacological testing and optimization, and will select the best lead compounds for testing in animal tumor models and advanced pre-clinical evaluation that will be needed before clinical trials can begin. If successful, this work would have an enormous impact on treating men with recurrent, high-grade prostate cancer.
Links:
Last updated Friday, March 7, 2025














