Prostate Cancer
Identifying and targeting enzalutamide resistance mechanisms in prostate cancer



Posted May 20, 2016
Dr. James Korkola and Dr. Joshi Alumkal, Oregon Health & Science University
 Castration-resistant prostate cancer (CRPC) is the lethal form of the disease that has become resistant to first line androgen-deprivation therapy. Cancer cells have been shown to adapt to androgen deprivation and continue activating the androgen receptor (AR) by developing the ability to synthesize their own androgens. The novel AR antagonist drug, enzalutamide, was developed to specifically target this androgen synthesis and activation mechanism. But even after initial success with enzalutamide treatment, resistance eventually occurs, so there is a great need to develop treatment options for enzalutamide-resistant prostate cancer patients. Recently, several groups identified a specific mutation (F877L) in the AR binding domain responsible for enzalutamide resistance or agonism of AR function in some cases, although the mechanisms that contribute to the agonist switch were not fully understood.
Castration-resistant prostate cancer (CRPC) is the lethal form of the disease that has become resistant to first line androgen-deprivation therapy. Cancer cells have been shown to adapt to androgen deprivation and continue activating the androgen receptor (AR) by developing the ability to synthesize their own androgens. The novel AR antagonist drug, enzalutamide, was developed to specifically target this androgen synthesis and activation mechanism. But even after initial success with enzalutamide treatment, resistance eventually occurs, so there is a great need to develop treatment options for enzalutamide-resistant prostate cancer patients. Recently, several groups identified a specific mutation (F877L) in the AR binding domain responsible for enzalutamide resistance or agonism of AR function in some cases, although the mechanisms that contribute to the agonist switch were not fully understood.
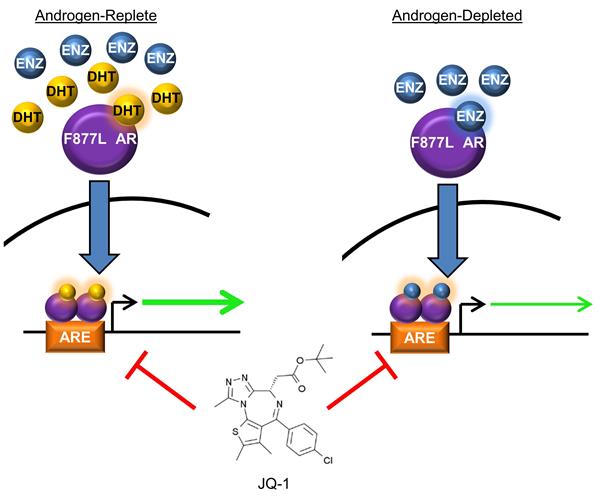
In studies funded through a FY12 Synergistic Idea Development Award, Drs. Korkola and Alumkal and their team from Oregon Health & Science University (OHSU) used a cellular model of acquired enzalutamide resistance to identify molecular mechanisms that contribute to enzalutamide agonism of mutant F877L AR. They determined that enzalutamide treatment of these cells led to the greatest agonistic effects when these cells were grown in the absence of androgen. This suggests that the androgen content of the cell may determine whether enzalutamide has the capacity to activate mutant F877L AR. Further, they demonstrated that enzalutamide and dihydrotestosterone, another form of androgen, activate a similar set of genes that are comprised largely of wild-type AR target genes. They also found that BET bromodomain inhibition suppressed either androgen or enzalutamide activation of mutant F877L AR and blocked enzalutamide-induced growth of mutant F877L AR CRPC tumors implanted in castrated mice (figure 1). A manuscript describing these findings is in press at Onctotarget.
The research team at OHSU is now translating these findings into the clinic by initiating a phase I clinical trial that will treat enzalutamide-resistant patients with the BET bromodomain inhibitor. This trial will enable them to verify mechanisms from their pre-clinical work and to determine if BET bromodomain inhibition is a promising strategy to block tumors harboring AR F877L mutations and other specific molecular alterations.
Links:
1. Coleman D, Hook KV, Lisac R, et al. Androgens interfere with enzalutamide agonism of mutant F876L androgen receptor Proc Am Assoc Ca Res. 2015;31(15):Abstr 3592.
2. Woodward J, King C, Coleman D, et al. Integrative genomic analysis to identify emergent enzalutamide resistance mechanisms in castration-resistant prostate cancer Proc Am Assoc Ca Res. 2015;30(23):Abstr 731.
3. Korkola JE, Liu M, Smith R, Liby T, Gray JW. NTRK1/TRKA as a therapeutic target in castration resistant prostate cancer. Proc Am Assoc Ca Res. 2016:Abstr 1347.
Last updated Thursday, December 5, 2024














