


Posted November 24, 2015
Howard Scher, M.D. and Michael Morris, M.D., Memorial Sloan Kettering Cancer Center
 The mission of the Prostate Cancer Clinical Trial Consortium (PCCTC) is to design, implement and complete hypothesis-driven early phase 1 and phase 2 clinical trials in prostate cancer, translating scientific discoveries to improved standards of care. Inherently linked to developing new drugs is the PCCTC's parallel commitment to developing more efficient ways of proving whether these drugs are beneficial to patients. The standards for multicenter clinical trial design and conduct developed under Department of Defense support have reduced the risk of abandoning an effective therapy, or moving an ineffective one forward. They also have reduced the time and number of patients needed to demonstrate that a drug works effectively, so that it can made available to patients sooner. As a direct result, the past several years have witnessed an unprecedented expansion of therapeutic options for men with metastatic castration-resistant prostate cancer (mCRPC). These new drugs are based on improved understanding of the underlying biology of the disease. However, unlocking their full therapeutic potential, optimizing their use, and accelerating future drug development requires better tools, called biomarkers, that have their own unique development path required for FDA approval. Biomarkers can be used to determine prognosis - how long a patient might live, assess response - is the treatment working, and/or to predict the likelihood a patient will respond to a given drug before it is administered thus guiding treatment selection and to determine if the drug is working after it is administered thus assessing response to the drug.
The mission of the Prostate Cancer Clinical Trial Consortium (PCCTC) is to design, implement and complete hypothesis-driven early phase 1 and phase 2 clinical trials in prostate cancer, translating scientific discoveries to improved standards of care. Inherently linked to developing new drugs is the PCCTC's parallel commitment to developing more efficient ways of proving whether these drugs are beneficial to patients. The standards for multicenter clinical trial design and conduct developed under Department of Defense support have reduced the risk of abandoning an effective therapy, or moving an ineffective one forward. They also have reduced the time and number of patients needed to demonstrate that a drug works effectively, so that it can made available to patients sooner. As a direct result, the past several years have witnessed an unprecedented expansion of therapeutic options for men with metastatic castration-resistant prostate cancer (mCRPC). These new drugs are based on improved understanding of the underlying biology of the disease. However, unlocking their full therapeutic potential, optimizing their use, and accelerating future drug development requires better tools, called biomarkers, that have their own unique development path required for FDA approval. Biomarkers can be used to determine prognosis - how long a patient might live, assess response - is the treatment working, and/or to predict the likelihood a patient will respond to a given drug before it is administered thus guiding treatment selection and to determine if the drug is working after it is administered thus assessing response to the drug.
Drs. Scher and Morris as well as their PCCTC colleagues, following the FDA's Critical Path Initiative, have developed the tools to reliably capture and characterize prostate cancer cells that are circulating in the blood stream (called circulating tumor cells, or CTCs), and novel ways of utilizing images of prostate cancer that has metastasized to distant sites in the body, particularly in the bone, where there have been no FDA approved standards to assess response. These biomarkers have been studied first in early phase trials of the PCCTC and later in large scale randomized phase 3 trials in which PCCTC played a leadership role. They have shown that a biomarker panel assessing CTC number and lactate dehydrogenase (LDH) levels in a patient's blood can be used as a response biomarker (surrogate) indicative of survival in a large phase III trial of abiraterone in men with mCRPC. In a similar trial, they demonstrated that a newly defined and standardized imaging outcome based on the number of new bone metastases that enables radiographic progression-free survival times to be determined accurately is also highly associated with survival. The bone progression outcome endpoint is now used in registrational trials that can lead to drug approvals. Thus this latest work has demonstrated that the CTC and imaging biomarkers are very useful in determining whether a patient is responding to treatment, and how long he might live.
The complexities of biomarker validation, analogous to phases of developing a drug, are underappreciated. Drugs are approved for indications, biomarkers are developed for contexts of use - the management decision that the biomarker result is intended to inform. With support from an FY12 PCRP Transformative Impact Award, Dr. Scher has assembled a multidisciplinary team from five premier medical centers, bringing together leaders in prostate cancer pathology, medical oncology, computational biology, and statistics. They have selected a set of biomarker tests for hormonal and biochemical signaling pathways that drive prostate cancer growth and metastasis including the androgen receptor (AR) and phosphatidylinositol-4,5-bisphosphate 3-kinase (PI3K) pathways. Their goal is to develop, validate, and obtain FDA approval for these biomarker assays to predict a patient's sensitivity to AR and PI3K inhibition.
The goal of the TIA Team is to use these assays as part of the eligibility criteria for protocols evaluating novel agents and separately, to guide the selection of approved agents that target these pathways. Successful deployment of such assays would allow patients to be given drugs that are most likely to benefit them and to avoid the toxicities of ineffective therapies. Identifying the patient population that would receive the highest benefit from a specific treatment will also accelerate drug approvals and the ability to use them in clinical practice to maximize patient benefit. The intention is to enable the practice of precision medicine where the choice of treatment is based on the real-time biologic profile of an individual patient's tumor.
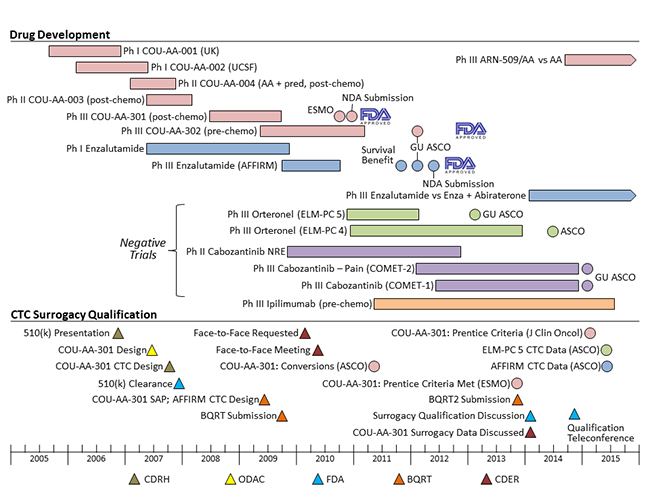
PCCTC-led codevelopment strategies accelerate the development of critically needed therapies and biomarkers
Publications:
Scher HI, Heller G, Molina A, Attard G, Danila DC, Jia X, Peng W, Sandhu SK,
Olmos D, Riisnaes R, McCormack R, Burzykowski T, Kheoh T, Fleisher M, Buyse M, de
Bono JS. Circulating tumor cell biomarker panel as an individual-level surrogate
for survival in metastatic castration-resistant prostate cancer. J Clin Oncol.
2015 Apr 20;33(12):1348-55. doi: 10.1200/JCO.2014.55.3487. Epub 2015 Mar 23.
PubMed PMID: 25800753; PubMed Central PMCID: PMC4397279.
Morris MJ, Molina A, Small EJ, de Bono JS, Logothetis CJ, Fizazi K, de Souza
P, Kantoff PW, Higano CS, Li J, Kheoh T, Larson SM, Matheny SL, Naini V,
Burzykowski T, Griffin TW, Scher HI, Ryan CJ. Radiographic progression-free
survival as a response biomarker in metastatic castration-resistant prostate
cancer: COU-AA-302 results. J Clin Oncol. 2015 Apr 20;33(12):1356-63. doi:
10.1200/JCO.2014.55.3875. Epub 2015 Jan 26. PubMed PMID: 25624432.
Links:














