


Posted March 26, 2015
Dr. Jun Luo, Johns Hopkins University
PC111969
 Prostate cancer is driven by male sex hormones (androgens) that mediate their effects by binding to the androgen receptor (AR). The standard initial treatment for men with metastatic prostate cancer consists of androgen deprivation therapy (ADT); however, the disease frequently stops responding to ADT and progresses to a state called castration-resistant prostate cancer (CRPC). Two new drugs, enzalutamide and abiraterone, were recently approved by the FDA and are now the most widely prescribed agents for CRPC. Unfortunately these therapies are very expensive, costing as much as $80,000 per year, and not every man with CRPC responds to these drugs. Having a test to measure drug response could help steer patients to these therapies when appropriate and also save non-responsive patients time and money from pursuing ineffective strategies.
Prostate cancer is driven by male sex hormones (androgens) that mediate their effects by binding to the androgen receptor (AR). The standard initial treatment for men with metastatic prostate cancer consists of androgen deprivation therapy (ADT); however, the disease frequently stops responding to ADT and progresses to a state called castration-resistant prostate cancer (CRPC). Two new drugs, enzalutamide and abiraterone, were recently approved by the FDA and are now the most widely prescribed agents for CRPC. Unfortunately these therapies are very expensive, costing as much as $80,000 per year, and not every man with CRPC responds to these drugs. Having a test to measure drug response could help steer patients to these therapies when appropriate and also save non-responsive patients time and money from pursuing ineffective strategies.
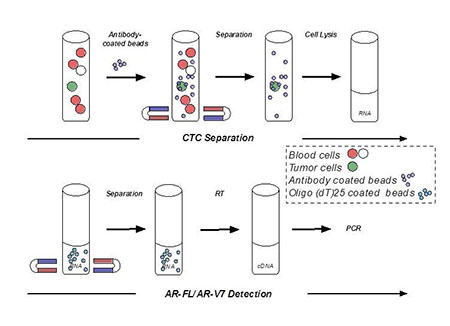
Preclinical studies suggested that an abnormal variant form of AR, called AR-V7, may mediate resistance to both enzalutamide and abiraterone by its ability to activate prostate cancer cells without requiring androgens. With this information in mind and through support from a FY11 PCRP Idea Development Award, Dr. Jun Luo and Co-Investigator Dr. Emmanuel Antonarakis at the Johns Hopkins University School of Medicine set out to develop and evaluate a blood-based assay to look for AR-V7 in circulating tumor cells (CTCs). A prospective study investigating the association of AR-V7 with resistance to enzalutamide and abiraterone found that men who were AR-V7-positive at the start of treatment did not derive any clinical benefit from either therapy. Conversely, the majority of patients without detectable AR-V7 in their blood did benefit significantly from treatment with these two drugs. Drs. Luo and Antonarakis also leveraged the resources of the PCRP supported Prostate Cancer Biorepository Network (PCBN) to analyze AR-V7 expression in metastatic tissue samples from a subset of men in the trial, and found concordance between AR-V7 expression in CTCs and solid metastases, leading them to conclude that detection of AR-V7 in patients with CRPC indicates resistance to enzalutamide and abiraterone.
Dr. Luo is now planning to simplify, standardize, and validate the AR-V7 test both analytically and clinically to assist in the transition to patient care for use in predicting therapeutic resistance. Validation would also enable the test to be incorporated into larger, prospective therapeutic trials of novel drugs to facilitate development of companion diagnostics. Thus, Dr. Luo's results show great promise for implementation of a non-invasive biomarker assay in both standard-of-care and clinical trial settings in the near future.
Publication:
Links:
Public and Technical Abstracts: Molecular Indicators of Castration-Resistant Prostate Cancer














