


Posted October 28, 2014
Elahe Mostaghel, MD PhD, Fred Hutchinson Cancer Research Center
 Ideally, all cases of prostate cancer would be prevented from ever occurring, or at the least aggressive forms of the disease would always be detected at the earliest stages to maximize treatment and survival. Unfortunately many cases of prostate cancer are detected in a more advanced disease state that ultimately progresses to lethal castration resistant prostate cancer. Docetaxel chemotherapy has been one of the few life-prolonging treatment options for these men, but recent FDA-approval of two new hormone therapy agents, abiraterone (ZYTIGA ©) and enzalutamide (XTANDI ©) has provided these patients with new treatment options. However, the optimal combination, sequence, and timing of these agents with docetaxel therapy have not been established. This is particularly relevant because one of the proposed anti-tumor activities of docetaxel (inhibition of the androgen receptor) could potentially synergize with the new hormone agents that also target this pathway.
Ideally, all cases of prostate cancer would be prevented from ever occurring, or at the least aggressive forms of the disease would always be detected at the earliest stages to maximize treatment and survival. Unfortunately many cases of prostate cancer are detected in a more advanced disease state that ultimately progresses to lethal castration resistant prostate cancer. Docetaxel chemotherapy has been one of the few life-prolonging treatment options for these men, but recent FDA-approval of two new hormone therapy agents, abiraterone (ZYTIGA ©) and enzalutamide (XTANDI ©) has provided these patients with new treatment options. However, the optimal combination, sequence, and timing of these agents with docetaxel therapy have not been established. This is particularly relevant because one of the proposed anti-tumor activities of docetaxel (inhibition of the androgen receptor) could potentially synergize with the new hormone agents that also target this pathway.
With this in mind, a team of researchers at Fred Hutchinson Cancer Research Center led by Dr. Elahe Mostaghel under the DoD PCRP Exploration-Hypothesis Development Award mechanism forged ahead to test the hypothesis that these new, more effective hormone therapy agents would synergize with and enhance cancer sensitivity to taxane treatments, such as docetaxel. To answer this question the team evaluated the impact of docetaxel and abiraterone on tumor growth of human derived castration resistant prostate cancer xenografts implanted in mouse models.
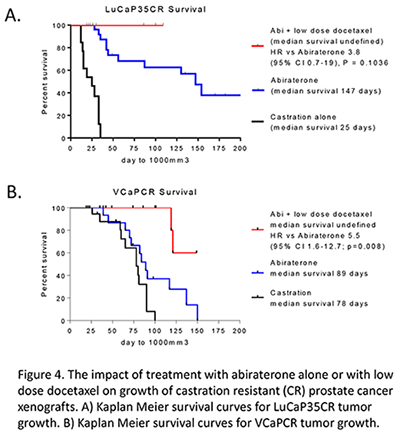
Dr. Mostaghel found no evidence that pre-treatment of the mice with abiraterone, to suppress intra-tumor androgen levels and androgen receptor signaling, had any beneficial impact on sensitizing tumors to taxane therapy. Instead, their data suggested that treatment with abiraterone might decrease sensitivity to subsequent taxane therapy. This is consistent with early clinical reports showing that following abiraterone treatment men are essentially resistant to taxane-based chemotherapy. However, they also made the surprising discovery that a combination of abiraterone treatment with several cycles of low dose docetaxel (which by itself did not have significant anti-tumor activity) actually improved treatment efficacy over abiraterone therapy alone.
These findings represent an attractive compromise for patients whose disease does not yet require chemotherapy, or who are unwilling or unable to undergo full dose taxane chemotherapy. Clinical evaluation of abiraterone plus low dose taxane is anticipated to demonstrate an improved benefit over abiraterone treatment alone, while forgoing the toxicity of full dose taxane therapy.
References:
Links:














