


Posted January 28, 2014
Daniel Danila, M.D., Memorial Sloan-Kettering Cancer Center
 Many treatment options for prostate cancer produce responses in only a subgroup of patients, but there is currently no way to predict which treatment is best for which patient. A specific type of tumor cell shed from both primary and metastatic tumors into the blood, called circulating tumor cells (CTCs), retain the intrinsic properties of the tumor, making them ideal candidates to serve as molecular biomarkers of treatment response in an effort to better "personalize" medicine.
Many treatment options for prostate cancer produce responses in only a subgroup of patients, but there is currently no way to predict which treatment is best for which patient. A specific type of tumor cell shed from both primary and metastatic tumors into the blood, called circulating tumor cells (CTCs), retain the intrinsic properties of the tumor, making them ideal candidates to serve as molecular biomarkers of treatment response in an effort to better "personalize" medicine.
Stemming from the observation in abiraterone acetate (ZYTIGA®) trials that patients with metastatic castration-resistant prostate cancer responded in variable patterns ranging from dramatic to intermediate to resistance after prolonged therapy, Dr. Daniel Danila, a FY08 Physician Research Training Award recipient, and his mentor Dr. Howard Scher led a team at the Memorial Sloan-Kettering Cancer Center (MSKCC) to address the mechanism of resistance for patients treated with abiraterone acetate. Their approach included predicting the association between tumor sensitivity to abiraterone treatment and molecular biomarkers detected in the CTCs collected from patients with metastatic prostate cancer. Dr. Danila and his team were able to demonstrate not only the feasibility of molecular profiling CTCs as a surrogate for sampling tumor tissue, but also that a significant proportion of abiraterone-treated patients have alterations of the prostate-specific fusion protein called TMPRSS:ERG. Important for clinical translation of these results, they also developed standard procedures for sample processing to ensure the validity of CTC assays.
Drs. Danila and Scher are now working to assess the potential correlation between TMPRSS:ERG with other factors (PTEN, PI3K, genomic copy number variations) in determining tumor sensitivity to treatment. Furthermore, their team is working to validate these assays to ensure their robustness and reproducibility in order to optimize the chance to ultimately incorporate the biomarker results in medical decision making, after testing them in conjunction with the clinical development of new targeted prostate cancer therapies being studied at MSKCC and within the DoD PCRP-funded Prostate Cancer Clinical Trials Consortium. The concurrent development of these CTC assays with the development of novel androgen receptor targeting agents will enable clinicians to prospectively identify patients most likely to benefit from treatment, which will not only accelerate the development of new androgen receptor targeting agents, but will ultimately improve patient outcomes from prostate cancer treatment.
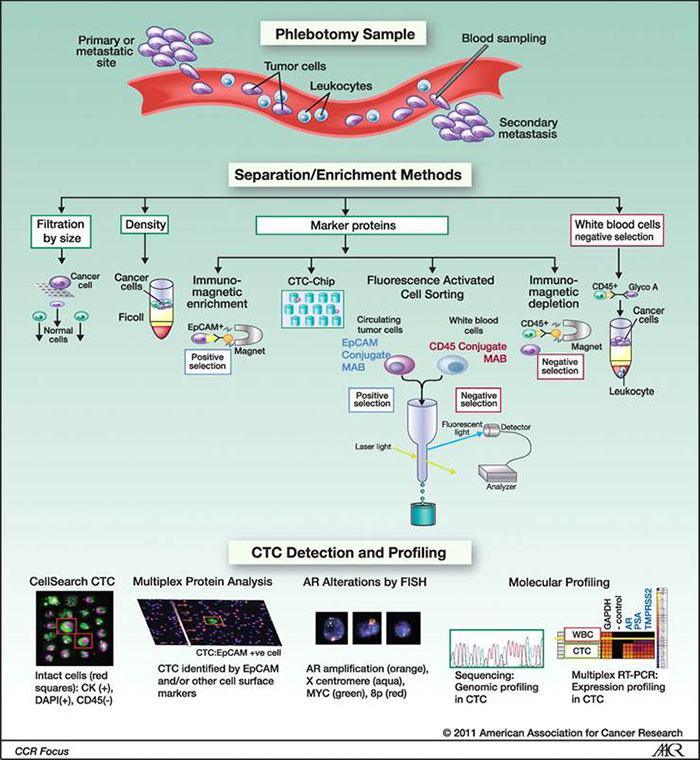
CTCs in patients with prostate cancer. Sampled by phlebotomy at the time when treatment is being considered, CTCs have the potential to provide tumor material for molecular profiling for biomarkers informative of tumor sensitivity to the targeted therapy being considered.
Publications:
Danila DC, Anand A, Sung CC, et al. 2011. TMPRSS2-ERG status in circulating tumor cells as a predictive biomarker of sensitivity in castration-resistant prostate cancer patients treated with abiraterone acetate. Eur Urol 60(5):897-904.
Danila DC, Fleisher M, and Scher HI. 2011. Circulating tumor cells as biomarkers in prostate cancer. Clin Cancer Res 17(12):3903-3912.
Links:














