


Posted April 30, 2013
Christopher Kloss, Weill Medical College of Cornell University
 Invasion of the body by harmful foreign bodies, such as bacteria and viruses, triggers an immune response that signals an attack and removal of the "non-self" invader. Cancerous cells can also be invaders of the body, but because of their origination from normal cells, the same immune response is not triggered, thereby allowing cancer cells to grow unchecked and eventually metastasize. Researchers believe that harnessing the ability of our immune system to specifically recognize and remove cancer cells would serve as a very potent therapy for prostate cancer patients. Under the mentorship of Dr. Michel Sadelain at Memorial Sloan Kettering Cancer Center, Christopher Kloss, a PhD student at Weill Medical College of Cornell University and a Predoctoral FY10 Prostate Cancer Training Award recipient, has been investigating a new method for engineering a patient's own T cells to target their prostate cancer.
Invasion of the body by harmful foreign bodies, such as bacteria and viruses, triggers an immune response that signals an attack and removal of the "non-self" invader. Cancerous cells can also be invaders of the body, but because of their origination from normal cells, the same immune response is not triggered, thereby allowing cancer cells to grow unchecked and eventually metastasize. Researchers believe that harnessing the ability of our immune system to specifically recognize and remove cancer cells would serve as a very potent therapy for prostate cancer patients. Under the mentorship of Dr. Michel Sadelain at Memorial Sloan Kettering Cancer Center, Christopher Kloss, a PhD student at Weill Medical College of Cornell University and a Predoctoral FY10 Prostate Cancer Training Award recipient, has been investigating a new method for engineering a patient's own T cells to target their prostate cancer.
Clinical trials have already showed promise with the method of engineering T cells to express a single artificial receptor that targets the T cells to cancer cells; however, this method is still not specific enough to prevent the T cells from also targeting normal cells. To solve this problem, Mr. Kloss engineered human T cells to express two artificial receptors instead of just one; one that induces CD3zeta signaling and a second that induces CD28 and CD137 signaling. This allows the T cells to target two antigens associated with the cancerous cells for their elimination while sparing normal cells that might express neither or either protein alone. The results from this study were recently published in Nature Biotechnology.
The ultimate goal would be to translate these findings into clinical trials for patient benefit. Similar studies in other cancer types have cured some patients, so this type of therapy shows great promise for prostate cancer treatment. However, it will likely be a challenge to make personalized medicine a cost effective therapy for patients. Mr. Kloss' career goals therefore extend beyond prostate cancer research to finding ways to make these therapies a reality. He plans to pursue an MBA degree once he has obtained his PhD from Cornell University so that he can become a more integral part of facilitating mergers between business and academia to support the trials and infrastructure necessary to make these personalized therapies a reality for patients.
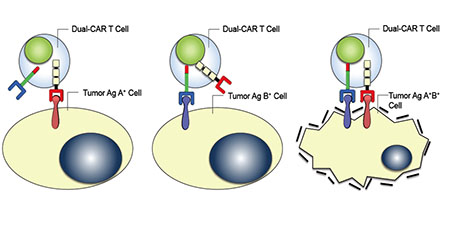
T cells with dual artificial receptors (chimeric antigen receptors, or CARs) are shown attacking other cells. Only cells with both tumor antigens (A and B) are eliminated.
References:
Kloss CC, Condomines M, Cartellieri M, Bachmann M, Sadelain M. 2013. Combinatorial antigen recognition with balanced signaling promotes selective tumor eradication by engineered T cells. Nature Biotechnology 31:71-75.














