Multiple Sclerosis
Bioengineered Particles to Promote Regulatory T Cells and Modulate the Immune System in Transplantation and Multiple Sclerosis



Posted August 10, 2023
Giorgio Raimondi, Ph.D., Jordan Green, Ph.D., Jamie Spangler, Ph.D. Johns Hopkins University
 Jamie Spangler, Ph.D.
Jamie Spangler, Ph.D.
(Photo Provided)
 Jordan Green, Ph.D.
Jordan Green, Ph.D.
(Photo Provided)
 Giorgio Raimondi, Ph.D.
Giorgio Raimondi, Ph.D.
(Photo Provided)
The immune system is a complex network of organs, cells, and proteins responsible for helping the body fight infections. When a virus or bacteria enter the body, cells of the immune system recognize these germs as foreign invaders and quickly mount a response to destroy them. However, sometimes the immune system mistakenly recognizes healthy and normal cells as foreign and attacks them, a condition called autoimmunity. For example, in multiple sclerosis (MS) the immune system attacks the myelin sheath, a protective covering that helps nerves send signals to the brain. This causes irreversible nerve damage that can lead to severe mobility and pain issues, affecting 1 million individuals in the US.
At other times, such as with transplantation, it is desirable that the immune system accept foreign tissues or organs without attacking them, a process called tolerance. The body's natural response is to reject transplanted tissue, requiring transplant recipients to take medication to suppress the immune response throughout their lifetime, leaving them more vulnerable to infection and cancer, as well as medication-induced kidney disease. Reconstructive transplantation is a type of transplant that can restore form and function to patients who have sustained catastrophic injuries to their face and/or upper extremities. In this type of procedure, termed vascularized composite allotransplantation (VCA), a hand or face is transplanted from a deceased donor to the recipient, replacing like with like.
In fiscal year 2017 (FY17), the Reconstructive Transplant Research Program (RTRP) awarded Dr. Giorgio Raimondi with a Concept Award to develop biodegradable particles for the selective stimulation and expansion of graft-protective T regulatory cells, or Tregs. Tregs are a type of immune cell that are responsible for suppressing other cells of the immune system to prevent autoimmune disease and maintain self-tolerance. Dr. Raimondi and his team engineered a fusion protein, called an immunocytokine, that combined a Treg-stimulating agent, interleukin-2, with an antibody that makes the agent available to just Tregs. This immunocytokine demonstrated the ability to expand and activate mouse and human Tregs.1 These results laid the groundwork for developing a Treg-based immunotherapy for modulating the immune system to prevent transplant rejection or treat autoimmune diseases.
With the support of an FY20 Multiple Sclerosis Research Program (MSRP) Exploration - Hypothesis Development Award, Dr. Raimondi and his research team designed biodegradable microparticles to selectively target the expansion of myelin-specific Tregs. This was achieved by fabricating biodegradable tolerogenic microparticles (Tol-MPs) containing the Treg-targeting and -expanding immunocytokine previously developed, a major histocompatibility complex II molecule with a myelin peptide fragment, and the immunosuppressant drug rapamycin, to further suppress the local immune response (Figure 1).1
In a recent Scientific Advances publication, the team describes the results of testing the Tol-MPs in a mouse model of MS that is characterized by progressive paralysis affecting the tail and hindlimbs. Injected Tol-MPs demonstrated the ability to delay disease onset and reduce disease severity when administered prior to disease induction. When administered after disease induction and symptom onset, the Tol-MPs almost completely reversed MS-like symptoms, with 37.5% of mice displaying no symptoms and 62.5% of mice showing only a minor loss of mobility in the tail.2
More studies are needed to fully characterize the extent of the protective effect of the Tol-MPs as well as testing in a relapsing-remitting model of MS. For transplantation,
Taken together, the outcomes of these RTRP and MSRP awards support the future development of bioengineered particles as a scalable transformative platform technology that is amenable to the creation of powerful immune-modulatory strategies for transplantation, multiple sclerosis, and many other autoimmune diseases.
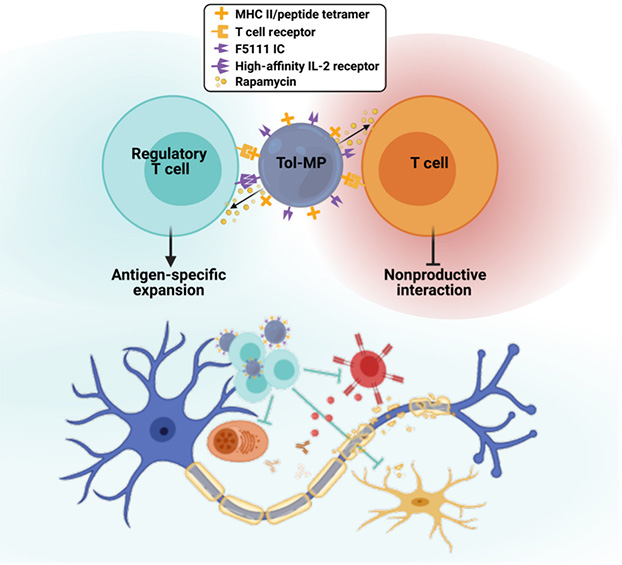 Fig. 1. Tol-MPs for Myelin-Specific Treg Induction.
Fig. 1. Tol-MPs for Myelin-Specific Treg Induction.
Rapamycin-loaded MPs are conjugated with tetrameric MHC class II/ peptide complexes and single-chain fusion IL-2/ anti-IL-2 (F5111) immunocytokines to generate tolerogenic MPs (Tol-MPs). Tol-MPs are designed to preferentially engage antigen-specific Tregs and promote their expansion. The immunocytokine blocks IL-2 signaling on non-Tregs that lack CD25, and the local release of rapamycin is intended to suppress pathogenic effector T cells that engage the Tol-MP. The expansion of myelin-specific Tregs can halt and reverse damage to the myelin sheath of nerves in the CNS to treat MS. Adapted from Rhodes KR et al.2
References:
1VanDyke D, Iglesias M, Tomala J, et al. 2022. Engineered human cytokine/antibody fusion proteins expand regulatory T cells and confer autoimmune disease protection.
2Rhodes KR, Tzeng SY, Iglesias M, et al. 2023. Bioengineered particles expand myelin-specific regulatory T cells and reverse autoreactivity in a mouse model of multiple sclerosis. Science Advances 9(22):eadd8693. https://doi.org/10.1126/sciadv.add8693
Links:
Public and Technical Abstracts: Localized and Inflammation-Responsive Immunotherapy of VCA Rejection
Last updated Wednesday, September 17, 2025
 An official website of the United States government
An official website of the United States government
 ) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
) or https:// means you've safely connected to the .mil website. Share sensitive information only on official, secure websites.
